GIZMO Reaction Energy-ALL ANSWERS CORRECT-VERIFIED A+
Document Content and Description Below
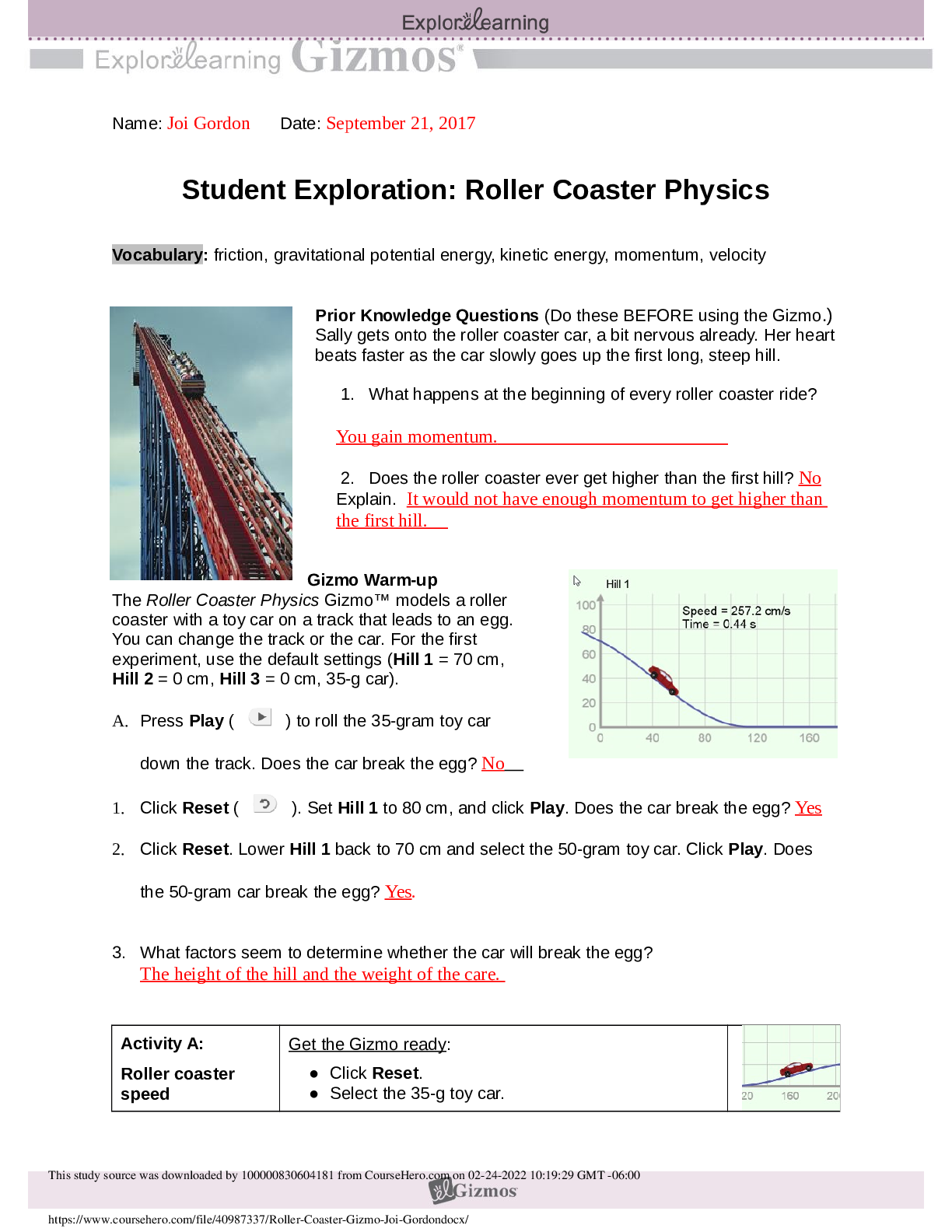
Student Exploration: Reaction Energy Vocabulary: calorimeter, chemical bond, endothermic, enthalpy, exothermic, Hess’s law Prior Knowledge Questions (Do these BEFORE using the Gizmo.) 1. Two magn... ets are stuck together. What might you have to do to get them to separate? Pull them apart 2. Suppose you held two magnets a short distance apart, then let go. What would happen? They would connect 3. Think about the magnets in terms of energy. In which case do you increase the potential energy of the magnets? In which case do you increase the kinetic energy of the magnets? Gizmo Warm-up Just like magnets, atoms of different elements are attracted together to form chemical bonds. Breaking these bonds requires energy. When a new bond forms, energy is released and temperatures rise. In the Reaction Energy Gizmo™, you will explore how the energy of chemical bonding relates to temperature changes that occur during chemical reactions. To begin, check that Reaction 1 and Forward are selected. In this reaction, hydrogen (H2) and oxygen (O2) react to form water (H2O). The reaction takes place inside a device called a calorimeter. Inside the calorimeter, a small chamber holds the reactants. The rest of the calorimeter is filled with water. 1. Click Play ( ). What happens? When I clicked play, all of the separate H2 and O2 molecules formed to create H2O. 2. How does the temperature change? The temperature is increased while the molecules are reacting to form water, and the increase in temperature slows once the molecules are all bonded to create the H2O molecules. The temperature raises from 21.0 Degrees Celsius to 26.0 Degrees Celsius while the molecules were bonding.Activity A: Energy of chemical bonds Get the Gizmo ready: Check that Reaction 1 and Forward are selected. Select the INVESTIGATION tab. Introduction: The heat energy stored in a chemical system is called the enthalpy (H) of the system. When atoms are joined by a chemical bond, energy must be added to pull them apart. This increases the enthalpy of the system. When a chemical bond forms, energy is released as shared electrons move into lower-energy orbitals. This causes the enthalpy to decrease. Question: How can you predict how much energy is released in a chemical reaction? 1. Predict: In the warm-up activity, you observed how the reaction inside the chamber affected the temperature of the surrounding water. Based on what happens to the surrounding water, do you think heat energy (enthalpy) is absorbed in the reaction or released? Explain. I think, based on what happened to the surrounding water, that enthalpy was released during the reaction and is therefore increased because energy was released as the shared electrons moved into lower-energy orbitals. 2. Observe: In the Gizmo, the energy required to break a chemical bond is modeled by placing a molecule into a set of mechanical claws. Place one of the hydrogen (H2) molecules between the claws, and press Break bond. A. What happens? Energy is absorbed to break apart the H2 bond. B. Look under the Energy absorbed column of the table. How much energy was required to break this bond? 436 kJ/mol was required to break the bond. Note: The energy is given here in units of kilojoules per mole (kJ/mol). This is the energy, in kilojoules, required to break all of the H–H bonds in one mole of H2 gas. C. Remove the hydrogen atoms from the claws and then break apart the other H–H molecule. What is the total energy absorbed so far? The total energy absorbed so far is 872 kJ/mol. 3. Measure: Notice that the oxygen atoms are connected by a double covalent bond. This is because the oxygen atoms share two pairs of electrons. Place the oxygen molecule in the claws and press Break bond. A. How much energy is required to break the first O–O bond? 349 kJ/mol is required to break the first O2 bond. B. Press Break bond. How much energy is needed to break both bonds? 495 kJ/mol is required to break both bonds.C. What is the total energy required to break up two moles of H2 molecules and one mole of O2 molecules? The total energy required to break up two moles of H2 molecules and one mole of O2 molecules is 1,367 kJ/mol. (Activity A continued on next page)Activity A (continued from previous page) 4. Create: Remove the two oxygen atoms from the claws. Now the claws disappear and you see a template for creating a water molecule. Drag an oxygen and a hydrogen atom into the template. (If necessary, use the Key on the right-hand side as a reference.) A. Click Create bond. What happens? Energy is released to create the bond. B. The “jiggling” animation you see represents the release of kinetic energy that occurs when a bond is formed. How much energy was released? 463 kJ/mol was released. C. Drag another hydrogen molecule into the template and click Create bond to make a water molecule. What is the total energy released so far? The total energy release so far is 926 kJ/mol. D. Drag the first water molecule away from the template, then use the Gizmo to create a second water molecule. What is the total energy released now? The total energy released now is 1,852 kJ/mol. 5. Calculate: Compare the energy absorbed in breaking up the molecules to the energy released when new bonds are formed. [Show More]
Last updated: 2 years ago
Preview 1 out of 13 pages

Buy this document to get the full access instantly
Instant Download Access after purchase
Buy NowInstant download
We Accept:

Reviews( 0 )
$11.00
Can't find what you want? Try our AI powered Search
Document information
Connected school, study & course
About the document
Uploaded On
Jun 25, 2021
Number of pages
13
Written in
Additional information
This document has been written for:
Uploaded
Jun 25, 2021
Downloads
0
Views
229