2021 solubility Temperature Gizmo (100% CORRECT ANSWERS)
Document Content and Description Below
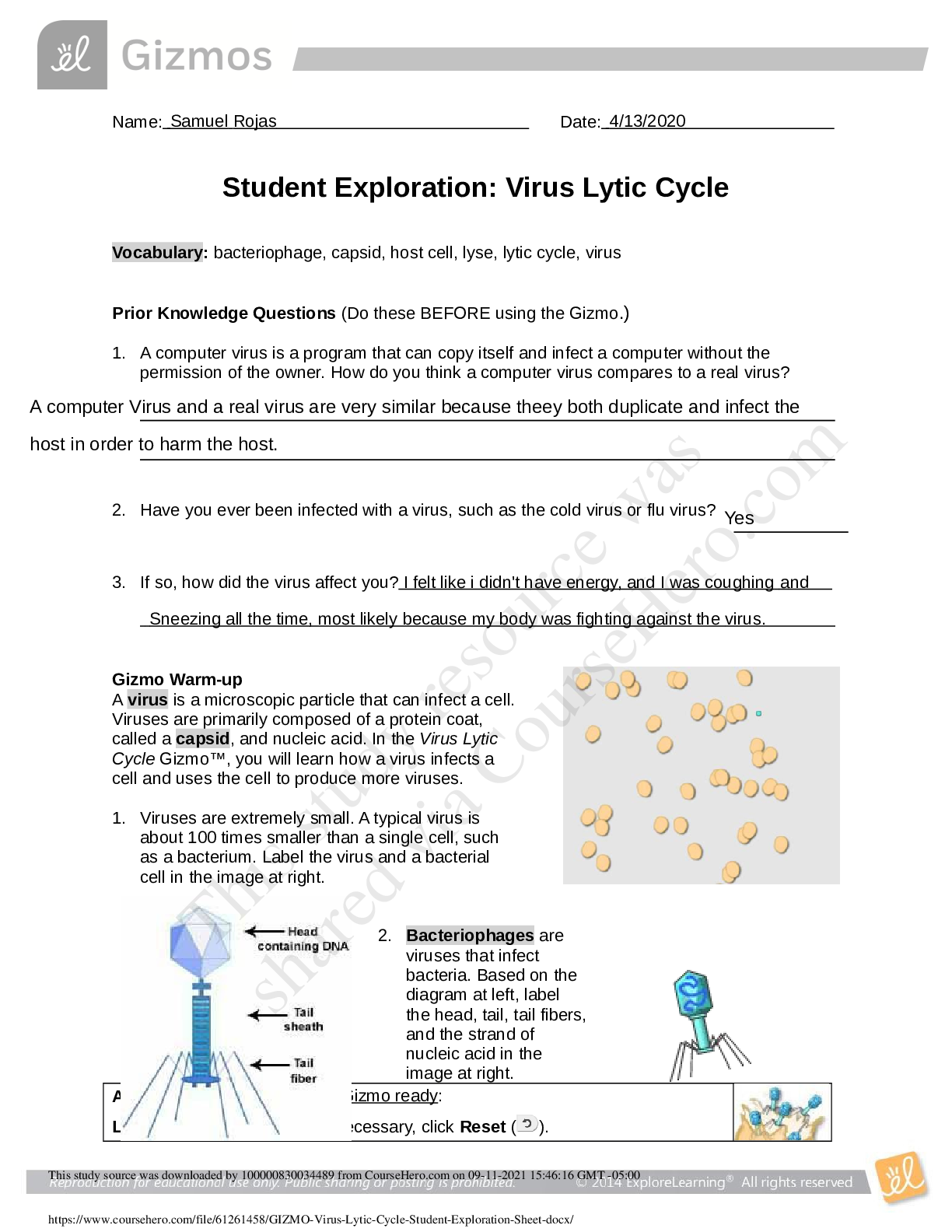
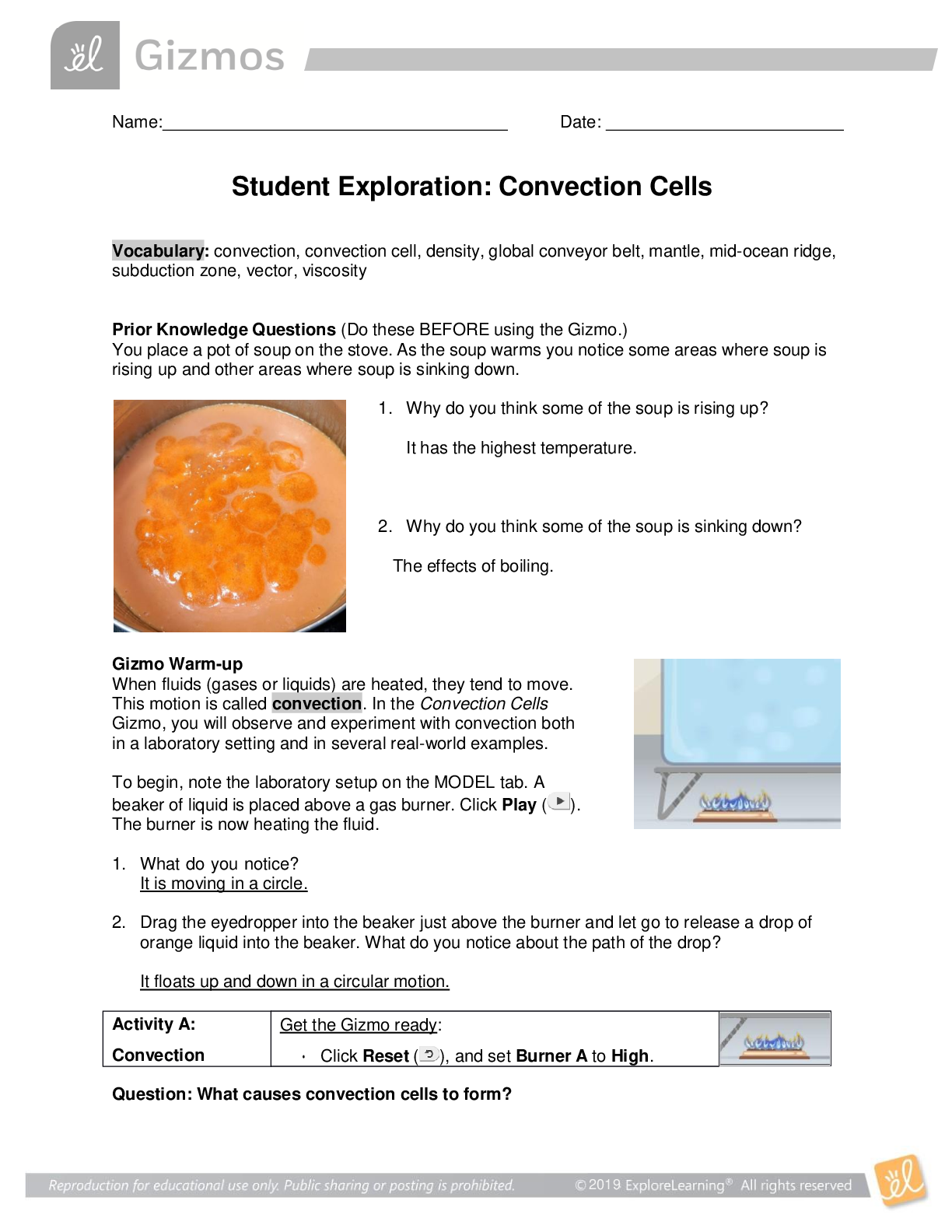
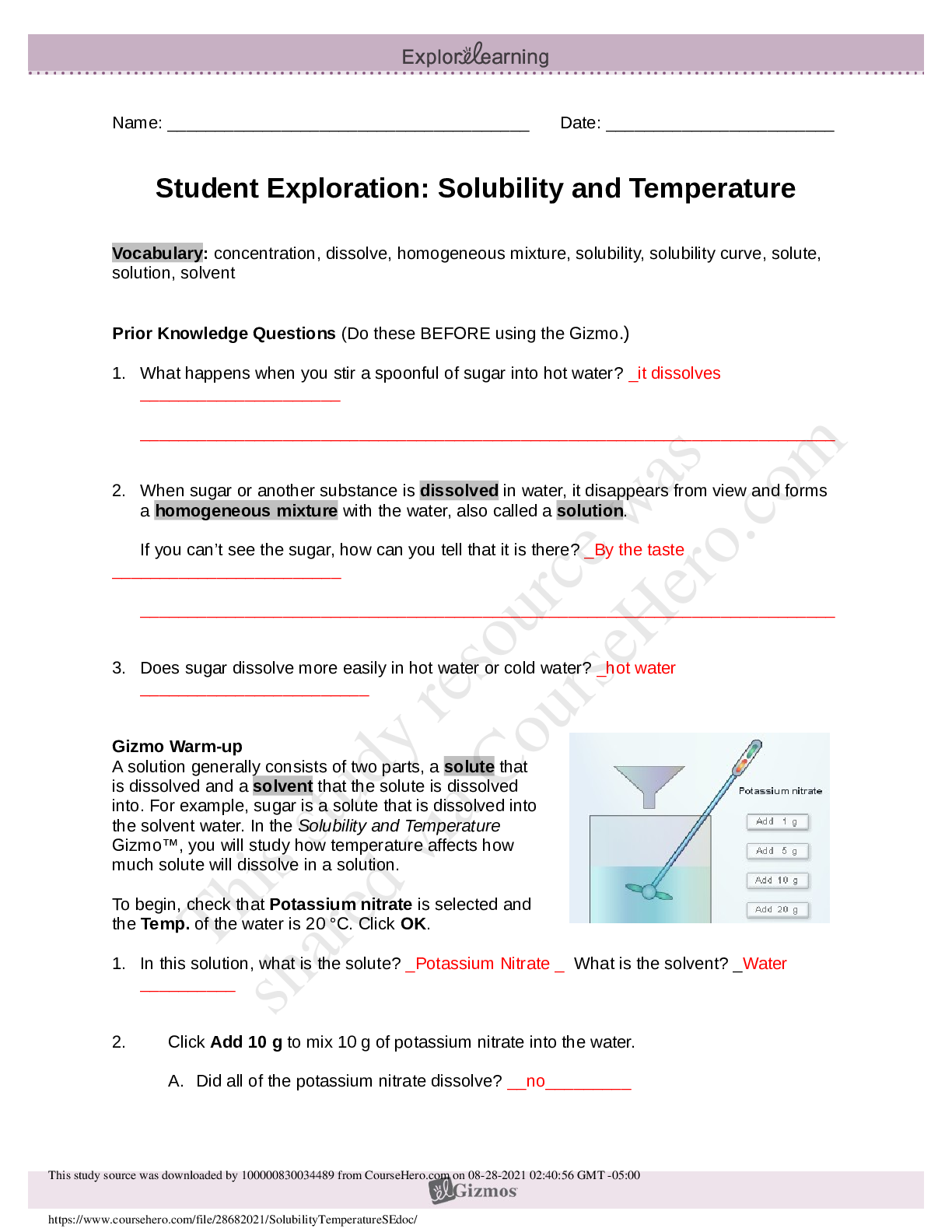
Solubility and Temperature NCVPS Chemistry Fall 2014 Vocabulary: concentration, dissolve, homogeneous mixture, solubility, solubility curve, solute, solution, solvent Prior Knowledge Questions (Do ... these BEFORE using the Gizmo.) 1. What happens when you stir a spoonful of sugar into hot water? It dissolves. 2. When sugar or another substance is dissolved in water, it disappears from view and forms a homogeneous mixture with the water, also called a solution. If you can’t see the sugar, how can you tell that it is there? You wouldn’t know unless you taste it since it dissolves. If the water is sweet then you’ll know sugar is in it. 3. Does sugar dissolve more easily in hot water or cold water? Hot water. Gizmo Warm-up A solution generally consists of two parts, a solute that is dissolved and a solvent that the solute is dissolved into. For example, sugar is a solute that is dissolved into the solvent water. In the Solubility and Temperature Gizmo™, you will study how temperature affects how much solute will dissolve in a solution. To begin, check that Potassium nitrate is selected and the Temp. of the water is 20 °C. Click OK. 1. In this solution, what is the solute? Potassium nitrate What is the solvent? Water 2. Click Add 10 g to mix 10 g of potassium nitrate into the water. A. Did all of the potassium nitrate dissolve? Yes B. How can you tell? You can’t see the solute (Potassium nitrate) because it’s none left over [Show More]
Last updated: 3 years ago
Preview 1 out of 7 pages

Buy this document to get the full access instantly
Instant Download Access after purchase
Buy NowInstant download
We Accept:

Reviews( 0 )
$12.00
Can't find what you want? Try our AI powered Search
Document information
Connected school, study & course
About the document
Uploaded On
Aug 28, 2021
Number of pages
7
Written in
All
Additional information
This document has been written for:
Uploaded
Aug 28, 2021
Downloads
0
Views
136