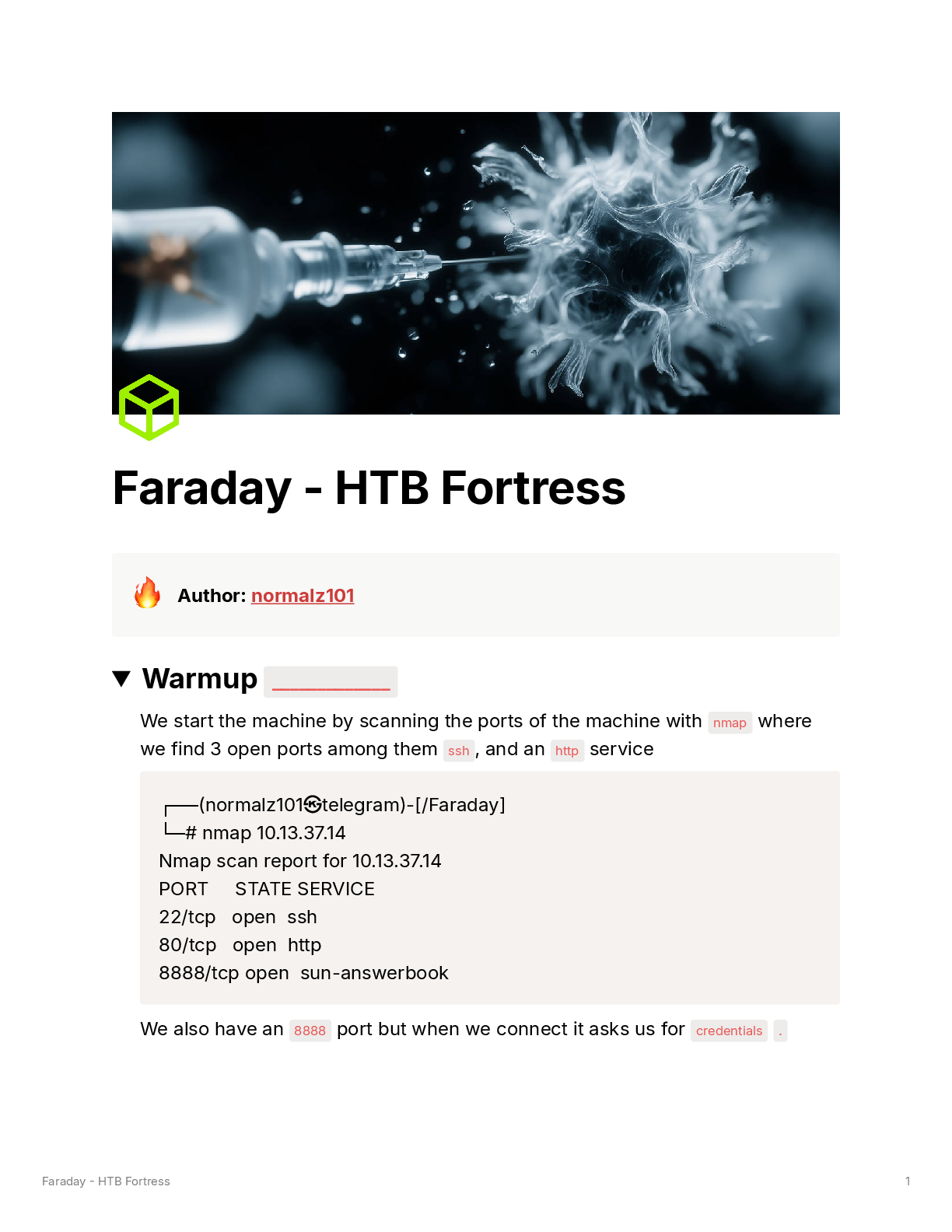
TABLE 23.2 Recommended Order of Treatment for Arrhythmias (Continued )
Order Agents Comments
Sustained
Ventricular
Tachycardia
First line Hemodynamically unstable patient: synchronized DCC
(100 J, biphasic).
Hemod
...
TABLE 23.2 Recommended Order of Treatment for Arrhythmias (Continued )
Order Agents Comments
Sustained
Ventricular
Tachycardia
First line Hemodynamically unstable patient: synchronized DCC
(100 J, biphasic).
Hemodynamically stable patient: IV procainamide,
IV amiodarone, or IV sotalol
Premedicate whenever possible when performing DCC.
Correct reversible causes.
Second line Hemodynamically stable patient: IV lidocaine; synchronized DCC should be considered if AAD therapy fails.
Once arrhythmia acutely terminated, patient should be
considered for ICD placement.
If patient refuses or is not a candidate for an ICD, PO
amiodarone can be considered.
If frequent shocks occur in patients with an ICD, PO amiodarone and β-blocker combination therapy or sotalol
monotherapy can be used.
Pulseless
Ventricular
Tachycardia/
Ventricular
Fibrillation
First line Start CPR, establish an airway, and deliver one shock
(biphasic defibrillator: 120–200 J; monophasic defibrillator: 360 J); immediately resume CPR for 2 min, then
check rhythm.
Correct reversible causes.
Second line If patient remains in pulseless VT/VF, deliver one shock
and then immediately resume CPR; if pulseless VT/VF
persists after at least one shock and CPR, give vasopressor therapy (epinephrine 1 mg IV push/IO every
3–5 min through pulseless VT/VF episode or vasopressin 40 units IV push/IO [to replace first or second dose
of epinephrine]) (give drugs during CPR; do not interrupt CPR to give drugs); immediately resume CPR for
2 min, then check pulse.
Third line If patient remains in pulseless VT/VF, deliver one shock
and then immediately resume CPR; if pulseless VT/
VF persists despite defibrillation, CPR and vasopressor therapy, consider AAD therapy (IV amiodarone;
lidocaine may be used if IV amiodarone unavailable)
(give drugs during CPR; do not interrupt CPR to give
drugs); consider IV magnesium sulfate if torsades de
pointes present; immediately resume CPR for 2 min,
then check pulse.
Bradycardia
First line Patient with stable bradycardia: Close observation
Patients with bradycardia and signs/symptoms of poor
perfusion (e.g., altered mental status, chest pain,
hypotension, shock): Immediately administer
IV atropine (0.5 mg every 3 to 5 min, up to 3 mg total
dose. If the atropine is ineffective, transcutaneous
pacing or sympathomimetic continuous infusion (dopamine or epinephrine) should be initiated.
Correct reversible causes.
Second line If drug therapy and transcutaneous pacing are ineffective, transvenous pacing should be utilized.
AV, atrioventricular; CPR, cardiopulmonary resuscitation; DCC, direct current cardioversion; ICD, implantable cardioverter-defibrillator; INR, International Normalized
Ratio; IV, intravenous; J, joules; LV, left ventricular; LVEF, left ventricular ejection fraction; MI, myocardial infarction; NSVT, nonsustained ventricular tachycardia; PO, oral;
PSVT, paroxysmal supraventricular tachycardia; PVC, premature ventricular contraction; TEE, transesophageal echocardiogram; VF, ventricular fibrillation; VT, ventricular
tachycardia
Arcangelo_Chap23.indd 322 10/8/2011 1:52:29 PM
CHAPTER 23 | ARRHYTHMIAS 323
in those patients with risk factors for thromboembolism, a
TEE-guided approach (see above) can be considered. Those
patients with risk factors for thromboembolism should also be
considered for at least 4 weeks of post-cardioversion anticoagulation therapy (Singer, et al., 2008).
If the practitioner decides to proceed with pharmacologic
cardioversion as the initial therapy, the selection of drug should
be based on the patient’s LV systolic function. Pharmacologic
cardioversion is most effective when initiated within 7 days
of the onset of AF. The AADs with proven efficacy during this time frame include dofetilide, flecainide, ibutilide,
propafenone, or amiodarone (oral or IV). The class Ia AADs,
disopyramide, procainamide, and quinidine have limited
efficacy or have been incompletely studied for this purpose.
Sotalol is not effective for converting AF to SR. Although the
use of single, oral loading doses of propafenone or flecainide
is effective in restoring SR, these drugs should only be used in
patients without underlying SHD. Ibutilide may also be considered in these patients. A patient’s ventricular rate should be
adequately controlled with AV nodal-blocking drugs prior to
administering a class Ic (or class Ia) AAD for cardioversion.
In patients with SHD, propafenone, flecainide, and ibutilide
should be avoided because of the increased risk of proarrhythmia. Instead, amiodarone or dofetilide should be primarily
used in this patient population. In patients with AF present
for more than 7 days, the only AADs with proven efficacy are
dofetilide, amiodarone (oral or IV), and ibutilide. The selection of AAD therapy during this time frame should again be
based on the presence of SHD (Fuster, et al., 2006).
If the practitioner does not wish to proceed with cardioversion, an initial management strategy of ventricular rate
control and anticoagulation is also reasonable. As previously
stated, this strategy, whereby the patient is left in AF, has been
shown to be an acceptable alternative to rhythm control for
the chronic management of AF. The selection of an oral drug
for chronic ventricular rate control is primarily based on the
patient’s LV systolic function. In patients with normal LV
systolic function (LVEF >40%), an oral b-blocker, diltiazem,
or verapamil is preferred over digoxin (Fuster, et al., 2006).
Digoxin can be added if adequate ventricular rate control
cannot be achieved with one of these drugs. In patients with
LVSD (LVEF of 40% or less), an oral b-blocker or digoxin is
preferred because these drugs can also concomitantly be used
to treat chronic HF. The nondihydropyridine CCBs should be
avoided in patients with LVSD because of their potent negative
inotropic effects. In patients with AF and stable HF symptoms
(NYHA class II or III), the b-blockers carvedilol, metoprolol
succinate, or bisoprolol should be used as first-line therapy
because of their documented survival benefits in patients
with HF (CIBIS II Investigators and Committees, 1999;
MERIT-HF Study Group, 1999; Packer, et al., 1996). Other
b-blockers should be avoided in these patients because their
effects on survival in HF are unknown. Digoxin should be used
as first-line therapy in patients with AF and decompensated
HF (NYHA class IV) because b-blocker therapy may exacerbate HF symptoms. For patients with normal or depressed
LV systolic function, oral amiodarone may also be considered
if adequate ventricular rate control cannot be achieved with the
use of b-blockers, nondihydropyridine CCBs, and/or digoxin
(Fuster, et al., 2006). In patients with persistent AF who have
no or acceptable symptoms and stable LV systolic function
(LVEF greater than 40%), the goal heart rate should be less than
110 beats/minute at rest (Van Gelder, et al., 2010; Wann, et al.,
2011). In patients with LVSD (LVEF of 40% or less), a stricter
heart rate goal (less than 80 beats/minute) should be considered
to minimize the potential harmful effects of a rapid heart rate
response on ventricular function (Wann, et al., 2011).
Assessing the patient’s risk of stroke becomes important
for selecting the most appropriate antithrombotic regimen.
The CHADS2 index is recommended for stroke risk stratification in patients with AF (Singer, et al., 2008). With this risk
index, patients with AF are given 2 points if they have a history of a previous stroke or transient ischemic attack and one
point each for being at least 75 years old, having hypertension,
having diabetes, or having congestive HF (CHADS2 is an
acronym for each of these risk factors). The points are added
up, and the total score is then used to determine the most
appropriate antithrombotic therapy for the patient. Patients
with a CHADS2 score of at least 2 are considered to be at high
risk for stroke and should receive warfarin (target INR: 2.5;
range: 2.0 to 3.0). Patients with a CHADS2 score of 1 are considered to be at intermediate risk for stroke and should receive
either warfarin (target INR: 2.5; range: 2.0 to 3.0) or aspirin
75–325 mg/d. However, because of its superior efficacy in
preventing stroke, the use of warfarin is suggested over that
of aspirin in this particular group of patients. Patients with a
CHADS2 score of 0 are considered to be at low risk for stroke
and should receive aspirin 75–325 mg/d. Dabigatran, an
oral direct thrombin inhibitor, can also be considered as an
alternative to warfarin for stroke prevention in patients with
paroxysmal or persistent AF and risk factors for stroke or systemic embolism (Wann, et al., 2011). Patients with prosthetic
heart valves, hemodynamically significant valvular disease, a
creatinine clearance less than 15 mL/minute, or advanced liver
disease are not appropriate candidates for dabigatran therapy.
Please refer to Chapter 54 for a further discussion of anticoagulation in AF.
Antithrombotic therapy should be considered for all
patients regardless of whether a rate-control or rhythm- control
strategy is initiated. In addition, antithrombotic therapy
should be continued if SR is restored because of the potential
for patients to have episodes of recurrent AF.
Third-Line Therapy
For those patients who remain symptomatic despite having
adequate ventricular rate control or for those patients in whom
adequate ventricular rate control cannot be achieved, it is reasonable to consider AAD therapy to maintain SR once they
have been converted to SR. The selection of an AAD to maintain SR is primarily based on the presence of SHD (Wann,
et al., 2011). (See Figure 23-2.) In patients without SHD,
any oral class Ia, Ic, or III AAD can be used to maintain SR.
Arcangelo_Chap23.indd 323 10/8/2011 1:52:29 PM
324 UNIT 4 | PHARMACOTHERAPY FOR CARDIOVASCULAR DISORDERS
However, dronedarone, flecainide, propafenone, or sotalol
should be considered as initial therapy in these patients because
of their less toxic adverse effect profiles. Amiodarone or
dofetilide can be used as alternative therapy if the patient fails
or does not tolerate one of these initial AADs. In patients with
any type of SHD, the class Ic AADs flecainide and propafenone
should be avoided. In these patients, the selection of AAD
therapy is based upon the type of SHD present. In patients
with LVSD (LVEF of 40% or less), either oral amiodarone or
dofetilide can be used. Both dronedarone and sotalol should be
avoided in patients with LVSD because of the risk of increased
mortality (dronedarone) or worsening HF (sotalol). In patients
with coronary artery disease, sotalol, dofetilide, or dronedarone can be used as initial therapy. In these patients, sotalol
and dronedarone should only be used if their LV systolic function is normal. Amiodarone can be considered as an alternative
therapy in these patients if these AADs are not tolerated. In
patients with significant LV hypertrophy, amiodarone is the
drug of choice.
For patients with permanent AF, a treatment strategy of
ventricular rate control and anticoagulation should be used
because the efficacy of AADs is extremely poor in this population. The oral drugs used for ventricular rate control are
discussed in the “Second-Line Therapy” section. Patients with
symptomatic episodes of recurrent AF who fail or do not tolerate at least one class I or III AAD may also be considered for
radiofrequency catheter ablation (Calkins, et al., 2007).
Paroxysmal Supraventricular Tachycardia (Due to
Atrioventricular Nodal Reentrant Tachycardia)
First-Line Therapy
Hemodynamically unstable PSVT requires first-line therapy of
synchronized DCC to restore SR and correct hemodynamic
compromise. Unless contraindicated, patients with mild to
moderate symptoms can be initially managed with vagal
maneuvers (e.g., unilateral carotid sinus massage, Valsalva
maneuver, facial immersion in ice water, and coughing).
Figure 23-3 illustrates an algorithm for the management of
PSVT due to AV nodal reentrant tachycardia.
Second-Line Therapy
If vagal maneuvers are unsuccessful or if PSVT recurs after successful vagal maneuvers, second-line therapy is AADs. The drug
of choice for PSVT is adenosine (American Heart Association,
2010). Clinical studies have shown that adenosine is as effective as IV verapamil in initial conversion of PSVT. Adenosine
does not produce hypotension to the degree that verapamil
does, and it has a shorter half-life. If a total of 30 mg of adenosine does not successfully terminate PSVT, further doses of this
agent are unlikely to be effective. Therefore, in patients with
persistent PSVT, other AADs will need to be used. In these
patients, IV diltiazem, verapamil, or a b-blocker can be used.
If PSVT continues despite these treatment measures, the use
of IV procainamide (LVEF >40%) or amiodarone (normal or
depressed LVEF) can also be considered.
Third-Line Therapy
Third-line therapy focuses on the management of chronic
PSVT. Chronic preventive therapy is usually necessary if the
patient has either frequent episodes of PSVT that require
therapeutic intervention or infrequent episodes of PSVT that
are accompanied by severe symptoms. Radiofrequency catheter ablation is considered first-line therapy for most of these
patients because of its effectiveness in preventing recurrence of
PSVT and its relatively low complication rate. Drug therapy
with oral diltiazem, verapamil, b-blockers, or digoxin can also
be considered if the patient is not a candidate for or refuses to
undergo radio-frequency catheter ablation.
Premature Ventricular Contractions
Occasional PVCs occur in most people and rarely compromise cardiac output or function. Correcting reversible causes
such as an electrolyte imbalance sometimes eliminates these
benign PVCs. Asymptomatic or minimally symptomatic
PVCs in patients without associated heart disease carry little
or no risk. PVCs in patients with heart disease were traditionally treated in the past. Decreasing the number and frequency
of PVCs was thought to diminish the risk of sudden cardiac
death. However, the results of the CAST showed that the use
of AADs to suppress asymptomatic PVCs in patients after
MI may increase mortality rates (CAST Investigators, 1989).
Therefore, if patients with SHD have symptomatic PVCs,
drug therapy should be limited to b-blockers. These agents
have been associated with a reduction in mortality and sudden
cardiac death in post-MI patients. These agents are also effective for suppressing symptomatic PVCs in patients without
SHD. Asymptomatic PVCs do not require treatment.
Nonsustained Ventricular Tachycardia
Ventricular tachycardia that spontaneously terminates within
30 seconds is known as nonsustained VT. Given the poor survival of patients who experience cardiac arrest, it is essential to
identify the most effective treatment strategies to prevent the
initial episode of sustained VT or sudden cardiac death from
occurring.
The presence of nonsustained VT in patients without
SHD is not associated with an increased risk of sudden cardiac death. Therefore, drug therapy is not necessary in these
patients if they are asymptomatic. However, if these patients
do become symptomatic, b-blocker therapy can be initiated.
Post-MI patients (especially those with LVSD) who develop
nonsustained VT are at increased risk for sudden cardiac
death. For these patients, the selection of therapy is based on
the patient’s LV systolic function. In post-MI patients with
an LVEF greater than 35%, drug therapy is not necessary to
treat the arrhythmia if they are asymptomatic. However, these
patients should still chronically receive a b-blocker specifically
to reduce mortality associated with the MI. b-blockers are also
effective if these patients develop significant symptoms associated with the nonsustained VT. In post-MI patients with
an LVEF of 35% or less, electrophysiologic testing is often
Arcangelo_Chap23.indd 324 10/8/2011 1:52:29 PM
CHAPTER 23 | ARRHYTHMIAS 325
performed when asymptomatic nonsustained VT occurs
(Moss, et al., 1996; Buxton, et al., 1999). If sustained VT or
VF is induced, an ICD is then recommended (Epstein, et al.,
2008). In these patients, implantation of the ICD should be
delayed until more than 40 days have elapsed since the MI
occurred. If sustained VT or VF is not induced, a b-blocker
or amiodarone can be initiated.
Sustained Ventricular Tachycardia
VT that persists for at least 30 seconds or that requires electrical or pharmacologic termination because of hemodynamic
instability is known as sustained VT. Since sustained VT can
degenerate into VF, the treatment goals are to terminate the
VT acutely and then prevent recurrence of the arrhythmia.
First-Line Therapy
If the patient is hemodynamically unstable (i.e., severe hypotension, syncope, HF, or angina), immediate synchronized DCC
is first-line therapy. If the patient is hemodynamically stable,
IV amiodarone, IV procainamide, or IV sotalol can be considered (American Heart Association, 2010). Lidocaine can be
used as alternative therapy. Synchronized DCC should be considered if AAD therapy fails.
Second-Line Therapy
Once the acute episode is terminated, measures should be
taken to prevent recurrent episodes of VT. Based on the results
of several trials, ICDs are clearly indicated as first-line therapy in patients with a history of sustained VT or VF (AVID
Investigators, 1997; Connolly, et al., 2000; Kuck, et al., 2000,
Epstein, et al., 2008). If the patient with an ICD experiences
frequent discharges because of recurrent ventricular arrhythmias or new-onset supraventricular arrhythmias (e.g., AF),
either amiodarone and a b-blocker or sotalol monotherapy
can be used. For the patient who refuses or is not a candidate
for an ICD, oral amiodarone should be used as an alternative
therapy.
Pulseless Ventricular Tachycardia/
Ventricular Fibrillation
The majority of cases of sudden cardiac death can be attributed
to VF. Sustained VT usually precedes VF and most commonly
occurs in patients with ischemic heart disease. VF is usually
not preceded by any symptoms and always results in a loss of
consciousness and eventually death if not treated. Immediate
treatment is essential in patients who develop VF or pulseless
VT, since survival is reduced by 10% for every minute that the
patient remains in the arrhythmia. It is imperative to identify
and correct any potential reversible causes for the arrhythmia.
For administration of drug therapy during an episode of
pulseless VT/VF, while IV access is preferred, the guidelines
recommend the intraosseous (IO) route as an alternative if
IV access cannot be established (American Heart Association,
2010). IO access can be used not only for administration of
drugs and fluids, but also for obtaining blood for laboratory
monitoring. If neither IV nor IO access can be established, the
endotracheal route can then be used for the administration of
only certain agents (i.e., atropine, lidocaine, epinephrine, and
vasopressin). Figure 23-4 illustrates an algorithm for the management of pulseless VT/VF.
First-Line Therapy
In patients with pulseless VT/VF, high-quality cardiopulmonary resuscitation (CPR) should be immediately initiated
until a defibrillator or automated external defibrillator (AED)
arrives (American Heart Association, 2010). High-quality
CPR is considered to be delivery of at least 100 compressions
per minute, with the depth of chest compressions being at
least 2 inches. Each cycle of CPR involves delivering 30 chest
compressions followed by two breaths. If a defibrillator or
AED is not readily available, hands-only CPR (compressions
only; no ventilations) should be provided if the bystander possesses no CPR training or is trained but lacks confidence in
providing effective CPR with rescue breaths. If the bystander
possesses CPR training and is confident in their ability to provide effective CPR with rescue breaths, conventional cycles of
CPR (30 chest compressions followed by two breaths) should
be delivered until a defibrillator or AED becomes available.
Once an advanced airway (e.g., endotracheal tube) is placed,
chest compressions should be delivered continuously at a rate
of 100 compressions per minute without pausing for ventilation (should be provided by a separate individual at a rate of
one breath every 6 to 8 seconds). Once a defibrillator or AED
arrives, defibrillation should be administered immediately.
With regard to defibrillation, delivery of only one shock
at a time is recommended in patients with pulseless VT/VF
to minimize interruptions in chest compressions (American
Heart Association, 2010). For biphasic defibrillators, the dose
of the shock is device-specific (usually 120 to 200 J); the
maximum dose available can be used for the initial shock if
the effective dose range of the defibrillator is unknown. This
dose or a higher dose can then be used for any subsequent
shocks that may be needed. After delivery of the initial shock
in patients with pulseless VT/VF, CPR should be immediately resumed and continued for 2 minutes, after which the
patient’s pulse and rhythm should be checked. Delaying
pulse and rhythm checks until after this period of CPR is
administered is intended to minimize interruptions in chest
compressions and increase the potential for success with defibrillation. If pulseless VT/VF persists, another shock should
be delivered at the appropriate dose, followed by 2 minutes
of CPR. This general sequence of resuscitation and defibrillation should be followed for as long as the patient remains in
pulseless VT/VF.
Second-Line Therapy
If pulseless VT/VF persists after delivery of at least one shock
and CPR, vasopressor therapy with either epinephrine or
vasopressin should be initiated (American Heart Association,
2010). One dose of vasopressin may be given to replace either
the first or second dose of epinephrine. Vasopressin’s half-life
Arcangelo_Chap23.indd 325 10/8/2011 1:52:29 PM
326 UNIT 4 | PHARMACOTHERAPY FOR CARDIOVASCULAR DISORDERS
of approximately 10 to 20 minutes is considerably longer than
the 3- to 5-minute half-life of epinephrine, which suggests that
its vasopressor effects may be more sustained than those of epinephrine during cardiac arrest. Unlike epinephrine, vasopressin also maintains its vasoconstrictive effects under acidotic
and hypoxic conditions, which suggests that this agent may
continue to work during prolonged cardiac arrest situations.
The recommended dosage of epinephrine for pulseless VT/
VF is 1 mg given IV push/IO every 3 to 5 minutes throughout the duration of the pulseless VT/VF episode. The recommended dosage of vasopressin for pulseless VT/VF is 40 units
IV push/IO for one dose only.
Third-Line Therapy
If pulseless VT/VF persists despite the use of defibrillation, CPR, and vasopressor therapy, AAD therapy can be
considered. IV amiodarone is recommended as first-line AAD
therapy for the treatment of pulseless VT/VF (American Heart
Association, 2010). This agent has been shown to be safe and
effective in the management of both in-hospital and outof-hospital pulseless VT/VF (Dorian, et al., 2002; Kudenchuck,
et al., 1999). Compared to lidocaine, IV amiodarone has been
associated with a significantly higher rate of survival to hospital
admission in patients with out-of-hospital cardiac arrest due to VF
(Dorian, et al., 2002). Therefore, lidocaine should only be considered for the treatment of pulseless VT/VF if IV amiodarone is not
available (American Heart Association, 2010). IV procainamide
is no longer recommended for pulseless VT/VF. IV magnesium
sulfate can be considered if TdP is present or suspected.
If the patient is resuscitated from the pulseless VT/VF episode, measures should be taken to prevent recurrent episodes
of cardiac arrest. Based on the results of several trials, ICDs are
clearly indicated as first-line therapy in patients with a history
of sustained VT or VF (AVID Investigators, 1997; Connolly,
et al., 2000; Kuck, et al., 2000; Epstein, et al., 2008). If patients
with an ICD experience frequent discharges because of recurrent ventricular arrhythmias or new-onset supraventricular
arrhythmias (e.g., AF), either amiodarone and a b-blocker or
sotalol monotherapy can be used. For patients who refuse or
are not candidates for an ICD, oral amiodarone should be used
as an alternative therapy.
Bradycardia
If patients with bradycardia present with signs and symptoms
of adequate perfusion, only close observation is required. If
patients with bradycardia develop signs or symptoms of poor
perfusion (e.g., altered mental status, chest pain, hypotension, shock), IV atropine (0.5 mg every 3 to 5 minutes, up to
3 mg total dose) should be immediately administered
(American Heart Association, 2010). If atropine is not effective, either transcutaneous pacing or a continuous infusion of
a sympathomimetic agent, such as dopamine (2 to 10 mcg/
kg/min) or epinephrine (2 to 10 mcg/min) (i.e., dopamine
or epinephrine) should be initiated. If symptomatic bradycardia persists despite any of these measures, transvenous pacing
should be utilized. Figure 23-5 illustrates an algorithm for the
management of bradycardia.
Special Population Considerations
Pediatric
The epidemiology of arrhythmias is different between adults
and children. Adults have arrhythmias primarily of a cardiac
origin, whereas children have arrhythmias primarily of a respiratory origin.
Tachyarrhythmias occasionally compromise infants and
young children. PSVT is the most common arrhythmia in young
children. It typically occurs during infancy or in children with
congenital heart disease. PSVT with ventricular rates exceeding
180 to 220 beats/minute can produce signs of shock. If signs
of shock appear, synchronized cardioversion or administration
of adenosine can be done in an emergency. Common causes of
PSVT in young children and infants are congenital heart disease (preoperative) such as Ebstein’s anomaly, transposition of
the great arteries, or a single ventricle. Postoperative PSVT also
can occur after atrial surgery for correction of congenital defects
of the heart. Other common causes of PSVT in children are
drugs such as sympathomimetics (cold medications, theophylline, b-agonists). WPW syndrome and hyperthyroidism also
can cause PSVT. Common causes of AF and atrial flutter in
children are intra-atrial surgery, Ebstein’s anomaly, heart disease
with dilated atria (aortic valve regurgitation), cardiomyopathy,
WPW syndrome, sick sinus syndrome, and myocarditis.
Bradycardia is a common arrhythmia in seriously ill
infants or children. It is usually associated with a fall in cardiac
output and is an ominous sign, suggesting that cardiac arrest is
imminent. The first-line therapy for this arrhythmia in infants
and young children is administration of oxygen, support respiration, epinephrine and, possibly, atropine.
Pulseless VT and VF are treated much the same way as
in adults; however, vasopressin is not currently recommended
for children (American Heart Association, 2010). The recommended dose of epinephrine for a child with pulseless VT/VF
is 0.01 mg/kg IV/IO, administered as 0.1 mL/kg of a 1:10,000
dilution. If IV/IO access cannot be established, epinephrine
can be administered endotracheally (0.1 mg/kg administered
as 0.1 mL/kg of a 1:1,000 dilution).
Geriatric
With aging, body fat increases, lean body tissue decreases, and
hepatic and renal system changes set the stage for potential overdosage and toxicity, particularly in the case of AADs. Similarly,
declining function affects the amount and dosage of the drug
prescribed as well as the occurrence of adverse effects. Cardiac
disease and chronic conditions such as HF exacerbate the decline
in organ function. Together, these factors can increase the risk of
an adverse effect from the AADs the practitioner prescribes.
For example, digoxin toxicity is relatively common in
elderly patients who are not receiving a reduced dosage to
accommodate for the reduced renal function. The practitioner
must always assess the patient’s baseline renal function (to identify abnormalities in the blood urea nitrogen and serum creatinine) and baseline hepatic function (to identify impairment in
liver function). These two tests are important in prescribing the
proper dosage of many of the AADs discussed in this chapter.
Arcangelo_Chap23.indd 326 10/8/2011 1:52:30 PM
CHAPTER 23 | ARRHYTHMIAS 327
Signs and symptoms of adverse effects of many drugs are
confusion, weakness, and lethargy. These signs and symptoms
are often attributed to senility or disease. Therefore, it is important for the practitioner to take a thorough drug history and to
document accurately the dosages and frequencies prescribed in
the patient record. If the practitioner merely attributes confusion to old age, the patient may continue to receive the drug
while actually experiencing drug toxicity. Furthermore, the
practitioner may add another drug to treat the complications
caused by the original AAD, compounding the issue of polypharmacy and excessive medication.
In elderly patients taking AADs, the practitioner must
be particularly alert to adverse effects from diuretics, digoxin,
sleeping aids, and nonprescription drugs.
AADs sometimes require accurate and timely dosing. If
an elderly patient forgets to take a dose or cannot remember
when he or she took the last dose, undermedication or overmedication may occur. This can be dangerous when AADs are
prescribed. Many of the elderly have multiple prescriptions,
even for the same medication, and therefore take an overdose
of the drug. Consequently, it is essential to review medications
with elderly patients and make sure they understand and can
follow a safe drug therapy regimen.
MONITORING PATIENT RESPONSE
The goals of AAD therapy are to restore SR and prevent
recurrences of the original arrhythmia or development of
new arrhythmias. Evaluating the outcomes of AAD therapy
requires the practitioner to schedule regular follow-up visits
after initial treatment of the arrhythmia. The outcomes to be
closely monitored include impulse generation and conduction
from the SA node to the AV node, time interval for conduction, heart rate within a normal range that is age-specific, and
patterns of AV and ventricular conduction.
Data to be monitored to evaluate therapeutic outcomes vary
from the simple to complex. The patient may monitor some of
them and needs to be taught the signs and symptoms to look for
and the expectations from the therapeutic regimen. Patients with
arrhythmias may be monitored on a regular or periodic basis with
12-lead ECG, 24-hour Holter monitors, electrophysiologic testing, monitoring of vital signs (blood pressure, pulse rate), echocardiograms for cardiac function, electrolytes, and serum drug levels.
In addition, the patient needs to self-monitor for symptoms such as lightheadedness, dizziness, syncopal episodes,
palpitations, chest pain, shortness of breath, or weight gain.
Other clinical outcomes to be monitored are those that affect
quality of life, such as activity tolerance, organ perfusion, cognitive function, fear, anxiety, and depression.
PATIENT EDUCATION
Drug Information
Included in the therapeutic plan for arrhythmia is patient education. Learning outcomes can be evaluated by monitoring
compliance with the medication regimen, recurrences of
arrhythmia, adverse effects, weight gain, blood pressure, heart
rate, and emergency department visits or hospitalizations.
AADs have narrow therapeutic windows. Toxicity is common at normal dosages. Consequently, patient education is
essential for providing maximal benefits and avoiding adverse
effects and accidental overdosing or underdosing.
The patient, family, and significant others should be taught
the basics, such as the name of the drug (both the generic and
trade name), the dose, the frequency and timing of the dose,
and the reason the drug is needed. This may avoid duplicate
prescribing and administration of AADs. The patient should
communicate, either verbally or in writing, the names and dosages of these drugs to all other health care providers and should
wear a medical identification device listing all medications. In
addition, the patient should inform his or her health care provider when any new prescription, over-the-counter, or complementary or alternative medications are started so that potential
drug interactions can be minimized or avoided.
The practitioner should provide written instructions for the
medication regimen. Providing instructions in large print and
simple language may be helpful to patients who have difficulty
with memory, hearing, or vision. Instructions should include
what to do when the patient misses a dose of medication, has an
adverse response to the medication, or wants to stop taking the
drug. If b-blockers are prescribed, the patient should be warned
that abrupt discontinuation may result in rebound angina, an
increased heart rate, and hypertension. The symptoms associated with these adverse effects also should be identified.
The practitioner can also teach the patient or caregiver
how to take blood pressure and pulse readings, how to interpret the readings, and how to recognize and respond to signs
and symptoms of hypotension, dizziness, chest pain, shortness of breath, peripheral edema, or palpitations. The patient
should take his or her weight each day and call the practitioner
if weight gain of over 2 pounds occurs. If the patient has difficulty learning these monitoring techniques or cannot perform them, he or she may need to schedule regular follow-up
appointments for monitoring. Patients with AF or atrial flutter
should know the signs and symptoms of a stroke.
In today’s health care environment, the insurance plan’s
pharmacy provider sometimes makes substitutions with generics or less expensive brands of medications. To prevent harmful drug effects, the patient needs to be aware of this practice
and should be cautioned not to change brands of the prescribed
AAD or anticoagulant without the approval of the practitioner.
An important teaching point from an ethical and legal
perspective is to warn the patient to avoid hazardous activities
such as driving, using electrical tools, climbing ladders, or any
activity that would put the patient or others in harm’s way
until the effects of the drug are demonstrated. Patients with
an ICD should refrain from driving for at least 6 months after
either implantation of the device or an appropriate discharge
from the device for a ventricular arrhythmia. Documentation
of patient teaching on risks, benefits, lifestyle modification, and
safety issues with AAD treatment should always be entered in
Arcangelo_Chap23.indd 327 10/8/2011 1:52:30 PM
328 UNIT 4 | PHARMACOTHERAPY FOR CARDIOVASCULAR DISORDERS
the patient’s record. Documenting a review of this information
on a follow-up visit aids health care providers who follow up
on the patient’s progress in the future.
Nutrition
Clear instructions should be given to avoid alcohol, excessive
salt intake, and caffeine during treatment for arrhythmias.
Many AADs may cause periods of hypotension resulting in
dizziness, or the dose of the drug may need to be regulated,
especially in the initial weeks.
Complementary and Alternative Medications
The practitioner must emphasize to the patient the importance
of reporting the use of any of these agents so that interactions
with AAD therapy can be minimized or avoided. While the
information regarding potential interactions between AADs
and specific complementary and alternative medications is
relatively sparse, there are a few notable interactions of which
practitioners should be aware. Patients taking AADs should
avoid licorice root. Licorice has mineralocorticoid effects,
which can promote hypokalemia. In patients taking digoxin,
the presence of hypokalemia may predispose the patient to
digoxin toxicity. In patients taking other AADs, the presence
of hypokalemia may promote the development of atrial or ventricular arrhythmias. In addition, certain licorice preparations
have been shown to cause a prolonged QT interval, which
may be additive in patients receiving class Ia or III AADs. This
interaction could lead to TdP. The use of Siberian ginseng or
oleander should also be avoided in patients receiving digoxin,
as digoxin toxicity may result. The use of St. John’s wort may
decrease digoxin concentrations; therefore, digoxin concentrations should be closely monitored when concomitant therapy
is used. St. John’s wort may also decrease plasma concentrations of amiodarone and dronedarone, which may predispose
the patient to arrhythmia recurrence. Consequently, the use
of St. John’s wort in patients receiving amiodarone or dronedarone should be avoided. Patients with a history of atrial or
ventricular arrhythmias should also be instructed to avoid the
use of any medication containing ephedra (e.g., Ma Huang)
because it can promote the development of arrhythmias.
BIBLIOGRAPHY
*Starred references are cited in the text.
*2010 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care science. (2010). Circulation,
122(Suppl 3), S1–S933.
*The Atrial Fibrillation Follow-up Investigation of Rhythm Management
(AFFIRM) Investigators. (2002). A comparison of rate control and
rhythm control in patients with atrial fibrillation. New England Journal of
Medicine, 347, 1825–1833.
*Anderson, J. L., Lutz, J. R., & Allison, S. B. (1983). Electrophysiologic and
antiarrhythmic effects of oral flecainide in patients with inducible ventricular tachycardia. Journal of the American College of Cardiology, 2, 105–114.
*Antiarrhythmics Versus Implantable Defibrillators (AVID) Investigators
(1997). A comparison of antiarrhythmic-drug therapy with implantable
defibrillators in patients resuscitated from near-fatal ventricular arrhythmias. New England Journal of Medicine, 337, 1576–1583.
*Bardy, G. H., Lee, K. L., Mark, D. B., et al. (2005). Amiodarone or an
implantable cardioverter-defibrillator for congestive heart failure. New
England Journal of Medicine, 352, 225–237.
*Buchanan, L. V., Turcotte, U. M., Kabell, G. G., & Gibson, J. K. (1993).
Antiarrhythmic and electrophysiologic effects of ibutilide in a chronic
canine model of atrial flutter. Journal of Cardiovascular Pharmacology, 33,
10–14.
*Buxton, A. E., Lee, K. L., Fisher, J. D., et al. (1999). A randomized study of
the prevention of sudden death in patients with coronary artery disease.
New England Journal of Medicine, 341, 1882–1890.
Cairns, J. A., Connolly, S. J., & Roberts, R. (1997). Randomized trial
of outcome after myocardial infarction in patients with frequent or
repetitive ventricular premature depolarizations: CAMIAT. Lancet, 349,
675–682.
*Calkins, H., Brugada, J., Packer, D.L., et al. (2007). HRS/EHRA/ECAS
expert consensus statement on catheter and surgical ablation of atrial
fibrillation: Recommendations for personnel, policy, and follow-up.
Heart Rhythm, 4, 816–861.
*Cardiac Suppression Trial Investigators. (1989). Preliminary report: Effect of
encainide and flecainide on mortality in a randomized trial of arrhythmia
suppression after myocardial infarction. New England Journal of Medicine,
321, 406–412.
*Carlsson, J., Miketic, S., Windeler, J., et al. (2004). Randomized trial of
rate-control versus rhythm-control in persistent atrial fibrillation: The
Strategies of Treatment of Atrial Fibrillation (STAF) study. Journal of the
American College of Cardiology, 41, 1690–1696.
*CIBIS II Investigators and Committees. (1999). The Cardiac Insufficiency
Bisoprolol Study II (CIBIS-II): A randomised trial. Lancet, 353, 9–13.
*Connolly, S., Gent, M., Roberts, R. S., et al. (2000). Canadian Implantable
Defibrillator Study (CIDS): A randomized trial of the implantable cardioverter defibrillator versus amiodarone. Circulation, 101, 1297–1302.
*Connolly, S. J., Dorian, P., Roberts, R. S., et al. (2006). Comparison of
beta-blockers, amiodarone plus beta-blockers, or sotalol for prevention
of shocks from implantable cardioverter defibrillators: The OPTIC study.
Journal of the American Medical Association, 295, 165–171.
Dager, W. E., Sanoski, C. A., Wiggins, B. S., & Tisdale, J. E. (2006).
Pharmacotherapy considerations in advanced cardiac life support.
Pharmacotherapy, 26, 1703–1729.
*Dorian, P., Cass, D., Schwartz, B., et al. (2002). Amiodarone as compared
with lidocaine for shock-resistant ventricular fibrillation. New England
Journal of Medicine, 346, 884–890.
*Duff, H. J., Roden, D., & Primm, R. K. (1983). Mexiletine in the treatment of resistant ventricular arrhythmias: Enhancement of efficacy and
reduction of dose-related side effects by combination with quinidine.
Circulation, 67, 1124–1128.
*Epstein, A. E., DiMarco, J. P., Ellenbogen, K. A., et al. (2008). ACC/AHA/
HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: A report of the American College of Cardiology/American Heart
Association Task Force on Practice Guidelines (Writing Committee to
Revise the ACC/AHA/NASPE 2002 Guideline Update for Implantation
of Cardiac Pacemakers and Antiarrhythmia Devices). Journal of the
American College of Cardiology, 51, e1–e62.
*Fuster, V., Rydén, L. E., Cannom, D. S., et al. (2006). ACC/AHA/ESC 2006
guidelines for the management of patients with atrial fibrillation: A report
of the American College of Cardiology/American Heart Association Task
Force on Practice Guidelines and the European Society of Cardiology
Committee for Practice Guidelines (Writing Committee to Revise the
2001 Guidelines for the Management of Atrial Fibrillation). Journal of the
American College of Cardiology, 48, e149–e246.
*Goldschlager, N., Epstein, A. E., Naccarelli, G., et al. (2007). A practical
guide for clinicians who treat patients with amiodarone. Heart Rhythm, 4,
1250–1259.
*Hohnloser, S. H., Crijns, H. J., van Eickels, M., et al. (2009). Effect of
dronedarone on cardiovascular events in atrial fibrillation. New England
Journal of Medicine, 360, 668–678.
Arcangelo_Chap23.indd 328 10/8/2011 1:52:30 PM
CHAPTER 23 | ARRHYTHMIAS 329
*Hohnloser, S. H., Kuck, K. H., & Lilienthal, J. (2000). Rhythm or rate
control in atrial fibrillation—Pharmacological Intervention in Atrial
Fibrillation (PIAF): A randomised trial. Lancet, 356, 1789–1794.
Julian, D. G., Camm, A. J., & Fragnin, G. (1997). Randomized trial of effect
of amiodarone on mortality inpatients with left ventricular dysfunction
after recent myocardial infarction: EMIAT. Lancet, 349, 667–674.
*Kennedy, H. L., Brooks, M. M., & Barker, A. H. (1994). Beta blocker
therapy in the Cardiac Arrhythmia Suppression Trial. American Journal
of Cardiology, 74, 674–680.
*Køber, L., Bloch-Thomsen, P. E., Møller, M., et al. (2000). Danish Investigations
of Arrhythmia and Mortality on Dofetilide (DIAMOND) Study Group.
Effect of dofetilide in patients with recent myocardial infarction and
left-ventricular dysfunction: A randomised trial. Lancet, 356, 2052–2058.
*Køber, L., Torp-Pedersen, C., McMurray, J. J., et al. (2008). Increased mortality after dronedarone therapy for severe heart failure. New England
Journal of Medicine, 358, 2678–2687.
*Kuck, K. H., Cappato, R., Siebels, J., et al. (2000). Randomized comparison
of antiarrhythmic drug therapy with implantable defibrillators in patients
resuscitated from cardiac arrest: The Cardiac Arrest Study Hamburg
(CASH). Circulation, 102, 748–754.
*Kudenchuck, P. J., Cobb, L. A., Copass, M. K., et al. (1999). Amiodarone for
resuscitation after out-of-hospital cardiac arrest due to ventricular fibrillation. New England Journal of Medicine, 341, 871–879.
*Lichstein, E., Morganroth, J., Harrist, R., & Hubble, M. S. (1983). Effect of
propranolol on ventricular arrhythmia: The beta blocker heart attack trial
experience. Circulation, 67 (Suppl. I), 5–10.
*MERIT-HF Study Group (1999). Effect of metoprolol CR/XL in chronic
heart failure: Metoprolol CR/XL Randomized Intervention Trial in
Congestive Heart Failure (MERIT-HF). Lancet, 353, 2001–2007.
*Miller, M. R., McNamara, R. L., Segal, J. B., et al. (2000). Efficacy of agents
for pharmacological conversion of atrial fibrillation and subsequent
maintenance of sinus rhythm: A meta-analysis of clinical trials. Journal of
Family Practice, 49, 1033–1046.
*Moss, A. J., Hall, W. J., Cannom, D. S., et al. (1996). Improved survival with an
implanted defibrillator in patients with coronary disease at high risk for ventricular arrhythmia. New England Journal of Medicine, 335, 1933–1940.
*Moss, A. J., Zareba, W., Hall, W. J., et al. (2002). Prophylactic implantation
of a defibrillator in patients with myocardial infarction and reduced ejection fraction. New England Journal of Medicine, 346, 877–883.
*Opolski, G., Torbicki, A., Kosior, D. A., et al. (2004). Rate control vs
rhythm control in patients with nonvalvular persistent atrial fibrillation:
The results of the Polish How to Treat Chronic Atrial Fibrillation (HOT
CAFE) Study. Chest, 126, 476–486.
*Oral, H., Souza, J. J., Michaud, G. F., et al. (1999). Facilitating transthoracic cardioversion of atrial fibrillation with ibutilide pretreatment.
New England Journal of Medicine, 340, 1849–1854.
*Pacifico, A., Hohnloser, S. H., Williams, J. H., et al. (1999). Prevention
of implantable-defibrillator shocks by treatment with sotalol: Sotalol
Implantable Cardioverter-Defibrillator Study Group. New England
Journal of Medicine, 340, 1855–1862.
*Packer, M., Bristow, M. R., Cohn, J. N., et al. (1996). The effect of carvedilol on morbidity and mortality in patients with chronic heart failure.
New England Journal of Medicine, 334, 1349–1355.
*Phillips, B. G., Gandhi, A. J., Sanoski, C. A., et al. (1997). Comparison
of intravenous diltiazem and verapamil for the acute treatment of atrial
fibrillation and flutter. Pharmacotherapy, 17, 1238–1245.
*Roy, D., Talajic, M., Dorian, P., et al. (2000). Amiodarone to prevent recurrence
of atrial fibrillation. New England Journal of Medicine, 342, 913–920.
*Roy, D., Talajic, M., Nattel, S., et al. (2008). Rhythm control versus rate
control for atrial fibrillation and heart failure. New England Journal of
Medicine, 358, 2667–2677.
*Sanoski, C. A., & Bauman, J. L. (2002). Clinical observations with the
amiodarone/warfarin interaction: Dosing relationships with long-term
therapy. Chest, 121, 19–23.
*Singer, D. E., Albers, G. W., Dalen, J. E., et al. (2008). Antithrombotic therapy
in atrial fibrillation: American College of Chest Physicians Evidence-Based
Clinical Practice Guidelines (8th ed.). Chest, 133, 546S–592S.
*Singh, B. N., Connolly, S. J., Crijns, H. J., et al. (2007). Dronedarone for
maintenance of sinus rhythm in atrial fibrillation or flutter. New England
Journal of Medicine, 357, 987–999.
*Singh, S., Zoble, R. G., Yellen, L., et al. (2000). Efficacy and safety of oral
dofetilide in converting to and maintaining sinus rhythm in patients
with chronic atrial fibrillation or atrial flutter: The Symptomatic Atrial
Fibrillation Investigative Research on Dofetilide (SAFIRE-D) Study.
Circulation, 102, 2385–2390.
*Slavik, R. S., Tisdale, J. E., & Borzak, S. (2001). Pharmacological conversion
of atrial fibrillation: A systematic review of available evidence. Progress in
Cardiovascular Diseases, 44, 121–152.
*Stambler, B. S., Wood, M. A., Ellenbogen, K. A., et al. (1996). Efficacy and
safety of repeated intravenous doses of ibutilide for rapid conversion of
atrial flutter or fibrillation. Circulation, 94, 1613–1621.
Stroke Prevention in Atrial Fibrillation Investigators. (1991). Stroke prevention in atrial fibrillation study: Final report. Circulation, 84, 527–539.
*Teo, K. K., Yusuf, S., & Furberg, C. D. (1993). Effects of prophylactic antiarrhythmic drug therapy in acute myocardial infarction: An overview of
results from randomized controlled trials. Journal of the American Medical
Association, 270, 1589–1595.
*Van Gelder, I. C., Groenveld, H. F., Crijns H. J. G. M., et al. (2010). Lenient
versus strict rate control in patients with atrial fibrillation. New England
Journal of Medicine, 362, 1363–1373.
*Van Gelder, I. C., Hagens, V. E., Bosker, H. A., et al. (2002). The Rate
Control Versus Electrical Cardioversion for Persistent Atrial Fibrillation
Study Group. A comparison of rate control and rhythm control in
patients with recurrent persistent atrial fibrillation. New England Journal
of Medicine, 347, 1834–1840.
*Vaughan Williams, E. M. (1984). A classification of antiarrhythmic actions
reassessed after a decade of new drugs. Journal of Clinical Pharmacology,
24, 129–147.
*Volgman, A. S., Winkel, E. M., Pinski, S. L., et al. (1998). Conversion
efficacy and safety of intravenous ibutilide compared with intravenous
procainamide in patients with atrial flutter or fibrillation. Journal of the
American College of Cardiology, 31, 1414–1419.
*Vukmir, R. B., & Stein, K. L. (1991). Torsades de pointes therapy with phenytoin. Annals of Emergency Medicine, 20, 198–200.
*Wann, L. S., Curtis, A. B., Ellenbogen, K. A., et al. (2011). 2011 ACCF/
AHA/HRS focused update on the management of patients with atrial
fibrillation (update on dabigatran): A report of the American College
of Cardiology Foundation/American Heart Association Task Force on
Practice Guidelines. Circulation, 123, 1144–1150.
*Wann, L. S., Curtis, A. B., January, C. T., et al. (2011). 2011 ACCF/AHA/
HRS focused update on the management of patients with atrial fibrillation (updating the 2006 guidelines): A report of the American College
of Cardiology Foundation/American Heart Association Task Force on
Practice Guidelines. Circulation, 123, 104–123.
*Wit, A. L., Rosen, M. R., & Hoffman, B. F. (1974). Electrophysiology
and pharmacology of cardiac arrhythmias: Relationship of normal and
abnormal electrical activity of cardiac fibers to the genesis of arrhythmias.
American Heart Journal, 88, 664–670, 798–806.
*Wyse, D. G., Kellen, J., & Rademaker, A. W. (1988). Prophylactic versus
selective lidocaine for early ventricular arrhythmias of myocardial infarction. Journal of the American College of Cardiology, 12, 507–513.
*Wyse, D. G., Waldo, A. L., DiMarco, J. P., et al. (2002). A comparison of
rate control and rhythm control in patients with atrial fibrillation. New
England Journal of Medicine, 347, 1825–1833.
Zipes, D. P., Camm, A. J., Borggrefe, M., et al. (2006). ACC/AHA/ESC 2006
guidelines for management of patients with ventricular arrhythmias and
the prevention of sudden cardiac death: A report of the American College
of Cardiology/American Heart Association Task Force and the European
Society of Cardiology Committee for Practice Guidelines (Writing
Committee to Develop Guidelines for Management of Patients with
Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death).
Journal of the American College of Cardiology, 48, e247–e346.
Arcangelo_Chap23.indd 329 10/8/2011 1:52:30 PM
Arcangelo_Chap23.indd 330 10/8/2011 1:52:30 PM
5 UNIT
Pharmacotherapy
for Respiratory
Disorders
Arcangelo_Chap24.indd 331 10/8/2011 1:52:50 PM
CHAPTER
332
Virginia P. Arcangelo
fall and spring, and coronavirus (10% to 15%), which is most
prevalent during the winter. The respiratory syncytial virus,
influenza virus, parainfluenza virus, and adenovirus are also
responsible, but the rhinovirus is the single most pervasive
cause of colds. The rhinovirus is a single-stranded ribonucleic
acid virus that replicates well at 95°F (35°C) or below but
poorly at 99 to 100°F (37.2 to 37.8°C), which is probably
why it causes URIs and not pneumonia.
Predisposition to viral infections can be attributed to many
factors, including frequent exposure to viral infectious agents;
in children, the age of the child; and the inability to resist
invading organisms because of allergies, malnutrition, immune
deficiencies, physical abnormalities, or other comorbid conditions. Some experts propose a relationship between the host
response to the virus and the production of cold symptoms.
Studies show that common colds are more frequent or more
severe in those under increased stress, probably as a result of
stress weakening the immune system.
PATHOPHYSIOLOGY
If the protective barriers of the upper respiratory tract (i.e., cough,
gag, and sneeze reflexes, lymph nodes, immunoglobulin [Ig] A
antibodies, and rich vasculature) fail, viral pathogens trigger an
acute inflammatory reaction with release of vasoactive mediators
and increased parasympathetic stimuli. This produces congestion and rhinorrhea. Rhinoviruses grow in the upper airway, and
attach and gain entry to host cells by binding to an intracellular
adhesion molecule. Infection begins in the adenoidal area and
spreads to the ciliated epithelium in the nose. Rhinoviruses are
hardy and remain infectious for at least 3 hours after drying on
hard surfaces such as telephones or countertops, but they do not
last as long on porous surfaces such as tissues.
Transmission of the virus has been attributed to three
methods: airborne transmission by small particles (droplets),
airborne transmission by large particles, and direct contact.
Large particle transmission is not efficient and requires prolonged exposure. The major means of transmission is by direct
contact from a donor’s nose to a donor’s hand, and from
there to the recipient’s hand and subsequently to the nose or
eye. Although conjunctival cells are not thought to harbor
Upper Respiratory Infections
Upper respiratory tract infections (URIs), including the common
cold and sinusitis, are some of the most common problems seen
in primary care. URIs are usually self-limiting, minor illnesses
that account for half or more of all acute illnesses. It is difficult
to differentiate the common cold from sinusitis or allergic rhinitis. (See Chapter 47.) URIs share common symptoms, such as
nasal discharge, nasal congestion, tenderness over the sinuses,
fever, headache, malaise, sore throat and myalgias, sneezing, a
full feeling around the eyes and ears, and coughing. Symptoms
may present individually or in combination, and it is difficult to
determine whether the cause is viral or bacterial.
URIs can progress to acute or chronic complications. In
children especially, URIs may progress to otitis media. In 5%
to 10% of cases, the viral or bacterial cause may travel, causing sinusitis and bronchitis. There is an enormous economic
burden associated with URIs.
COMMON COLD
Acute infectious rhinitis (coryza), or the common cold, is a viral
URI. One of the most common infections, it is self-limiting.
Coryza is an acute inflammation of the mucous membranes of the
respiratory passages, particularly of the nose, sinuses, and throat,
and is characterized by sneezing, rhinorrhea (watery nasal discharge), and coughing. The common cold has a short duration.
Approximately 100 million colds occur annually in the
United States, resulting in approximately 26 million days off from
school, 23 million absent days from work, 27 million visits to a
primary care provider, and 250 million days of restricted activities. Nearly $1 billion is spent on cold remedies and $1.5 billion
on analgesics. Adults average three colds per year. Children average six episodes per year, and the common cold is more common
in children who attend day care or preschool (where they are in
contact with other children and groups that may spread disease)
than in those who spend more time at home and have less contact
with crowds. Exposure to smoke is also a predisposing factor.
CAUSES
The pathogens most frequently associated with common colds
are rhinovirus (30% to 40% of cases), especially during the
24
Arcangelo_Chap24.indd 332 10/8/2011 1:52:50 PM
CHAPTER 24 | UPPER RESPIRATORY INFECTIONS 333
rhinovirus, it probably can be passed through the tear duct
into the nose. Incubation of the rhinovirus is 1 to 10 days.
Onset of signs and symptoms occurs 1 to 2 days after viral
infection, and they peak in approximately 2 to 4 days. The
virus may remain present for a week or longer after the onset of
symptoms. A cough may persist after other symptoms resolve.
DIAGNOSTIC CRITERIA
Diagnostic tests have no cost/benefit effect in diagnosing the
common cold. Symptoms consist primarily of clear nasal discharge, sneezing, nasal congestion, cough, low-grade fever (below
102°F [38.9°C]), scratchy or sore throat, mild aches, chills,
headache, watery eyes, tenderness around the eyes, full feeling
in the ears, and fatigue. In children, the presentation could also
include nasal blockage, fever with seizures, anorexia, vomiting,
diarrhea, and abdominal pain. Symptoms usually resolve in
approximately 1 week, but they may linger for 2 weeks.
INITIATING DRUG THERAPY
Mistreatment of the common cold by clinicians is common
for two reasons:
• It is difficult to determine whether the cause is viral or
bacterial.
• Patients often have preconceived notions and demand antibiotics for their URI even though it is simply the common
cold, which is caused by a virus.
There is no cure for the common cold. Treatment is geared
toward minimizing symptoms (Table 24.1).
Nonpharmacologic alternatives to treating the common
cold are the first line of treatment. For example, rest allows the
body to gain strength and be more effective in defending itself
against the pathogen. The body can then dictate the increase
in activities. An alternative to decongestants and expectorants
is increasing water or juice intake. This assists in liquefying
tenacious secretions, making expectoration easier, soothing
scratchy, sore throats, and relieving dry skin and lips. Saline
gargles also are effective for soothing sore throats.
Coughing caused by chest congestion can cause a muscular chest pain. Menthol rubs can soothe this ache and open
airways for some congestion relief. Menthol lozenges also have
been effective in soothing scratchy throats and clearing nasal
passages. Saline nasal flushes are also effective for clearing nasal
passages without the rebound side effect. Petrolatum-based
ointments for raw and macerated skin around the nose and
upper lip ease the drying effects of dehydration and the use of
multiple tissues. (See Table 24.1.)
Other measures, such as drinking chicken soup, taking
a hot shower, or using a room humidifier, may prove helpful.
TABLE 24.1 Alternative Therapies for Cold Symptoms
Symptoms Nonpharmacologic Pharmacologic Alternative Therapy
Any cold symptoms Eat proper diet, rest, drink fluids. Echinacea (prevention)
Zinc lozenges (decreased
duration of symptoms)
Rhinorrhea Use disposable paper tissues. Anticholinergic nasal spray
Nasal obstruction Decrease ingestion of milk
products.
Children: saline nose drops by bulb
syringe
Bayberry tea
Inhale warm, moist heat, such as
showers.
Apply topical decongestants.
Increase fluid intake. If nasal obstruction is still a problem
after 3 d, take oral decongestants
unless contraindicated by hypertension or coronary artery disease.
Serous otitis media or sensation of fullness in ears
Decongestants (oral)
Headache, sore throat,
malaise, myalgia, fever
Gargle with salt water, drink
plenty of fluids, suck on
menthol lozenges.
Nonsteroidal anti-inflammatory drugs Chaparral, aromatherapy
rubs, boneset
Chest congestion Drink fluids, have menthol rubs,
and humidify room air.
Expectorants
Sneezing and watery eyes Humidify room air. Two schools of thought: antihistamine
of choice, but critics say antihistamines not needed in treating colds,
especially in children
Cough Humidify room air. Antitussives, naproxen
Vicks VapoRub on the soles of feet
covered by socks at bedtime
Arcangelo_Chap24.indd 333 10/8/2011 1:52:50 PM
334 UNIT 5 | PHARMACOTHERAPY FOR RESPIRATORY DISORDERS
Inhaling warm, moist heat helps raise the temperature of the
nasal mucosa to at least 98.6°F (37°C), a temperature at which
the virus does not replicate so readily.
Applying Vicks VapoRub on the soles of the feet and then
putting on socks may help with a persistent night-time cough.
Goals of Drug Therapy
The main goals of treatment for the common cold are relief of
symptoms, reduction of the risk for complications, and prevention of spread to others (Box 24.1). Polypharmacy is often
used to treat intolerable symptoms.
Decongestants
Mechanism of Action
Decongestants are sympathomimetic agents that stimulate
alpha- and beta-adrenergic receptors, causing vasoconstriction
in the respiratory tract mucosa and thereby improving ventilation (Table 24.2). Decongestants come in topical or oral
preparations. Topical decongestants in the form of nasal sprays
slow ciliary motility and mucociliary clearance. Topical agents
have little systemic absorption. However, topical decongestants
should not be used for more than 3 days because prolonged use
can cause rhinitis medicamentosa (rebound congestion), which
is characterized by severe nasal edema, rebound congestion,
and increased discharge due to decreased receptor sensitivity.
Oral decongestants are frequently used and are sold over
the counter (OTC) alone or in combination with other drugs.
A common example of a combination preparation is an antihistamine and a decongestant. The most common oral decongestant is pseudoephedrine (Sudafed, others). Oral decongestants
have the same mode of action as topical agents but can cause
more systemic responses. Decongestants assist in clearing nasal
obstruction. Their use may be encouraged to prevent sinusitis
and eustachian tube blockage.
Contraindications
Decongestants are contraindicated in patients with narrowangle glaucoma, hypertension, and severe coronary artery
disease. Caution is recommended in patients with hyperthyroidism, diabetes, and prostatic hypertrophy (causes difficulty
with urination).
Adverse Events
Adverse events include increased blood pressure, increased
heart rate, palpitations, headache, dizziness, gastrointestinal
(GI) distress, and tremor. These reactions are especially seen at
doses above 210 mg.
Interactions
Decongestants interact with appetite suppressants, monoamine oxidase (MAO) inhibitors (hypertensive crisis), and
beta- adrenergic agents (bradycardia and hypertension).
Decongestants are less effective when taken with drugs that
acidify the urine and more effective when taken with drugs
that alkalize the urine.
Expectorants
One of the most important nondrug considerations in treating
coughs is discovering its cause, because the prolonged use of
OTC expectorants or other cough products may mask symptoms of a serious underlying disorder. The drug should not be
used for more than a week. If the cough persists, additional
measures may be investigated.
Mechanism of Action
Expectorants, including water, increase the output of respiratory tract fluid by decreasing the adhesiveness and surface
tension of the respiratory tract and by facilitating removal of
viscous mucous. (See Table 24.2.) The effect is noted within
1 to 2 hours.
Adverse Events
Adverse events include drowsiness, headache, and GI symptoms.
Antitussives
Cough is a frequent complaint of a person with an URI. Cough
can be stimulated from congestion or can occur as a result of
postnasal drip. There are many cough suppressants available,
but studies have shown minimal benefit with the common cold.
• Avoid touching face with hands.
• Wash hands frequently.
• Avoid people with colds or URIs.
• Avoid crowded areas during peak URI/flu season.
• Stop smoking.
• Avoid excessive intake of alcohol.
• Avoid second-hand smoke.
• Avoid excessive dry heat.
• Use disposable tissues and dispose of them properly.
• Obtain influenza vaccine, if recommended.
• Eat a nutritious diet.
• Get adequate rest and sleep.
• Avoid or reduce stress.
• Exercise regularly.
• Increase humidity in the house, especially during
winter months.
• Maintain good oral hygiene.
• Avoid certain environmental irritants and allergens
(dust, chemicals, smoke, and animal dander) when
possible.
• Use central ventilation fans/air conditioning with
microstatic air filters.
BOX 24.1
Preventing Upper
Respiratory Infections
Arcangelo_Chap24.indd 334 10/8/2011 1:52:50 PM
CHAPTER 24 | UPPER RESPIRATORY INFECTIONS 335
TABLE 24.2 Overview of Agents for Upper Respiratory Infections
Generic (Trade)
Name and Dosage Selected Adverse Events Contraindications Special Considerations
Decongestants
oxymetazoline hydrochloride
(Afrin) ≥6 y: 2–3 sprays bid 2–6 y
(use children’s spray): 2–3 sprays
bid
Palpitations, headaches These drugs may cause
rebound congestion.
Use only 2 to 3 days, then
switch to oral decongestants.
phenylephrine hydrochloride
(Neo-Synephrine)
Adults: 1 spray q3–4h as needed
Palpitations, headaches Not recommended for
children
These drugs may cause
rebound congestion.
Use only 2 to 3 days, then
switch to oral decongestants.
pseudoephedrine (Sudafed, Benylin
decongestant)
Adults: short acting: 60 mg q4–6h;
long acting: 120 mg q12h
Children 7–12 y: short acting: 30 mg
q4–6h
Children 3–6 y: 15 mg q4–6h
Palpitations, headaches,
increased blood pressure,
dizziness, GI upset, tremor
Hypertension, coronary
artery disease
Give at least 2 h before bedtime. Do not crush, break, or
chew tablets.
Expectorants
guaifenesin (Antitussin, Mucinex,
Robitussin, Uni-Tussin)
Adults: short acting: 200–400 mg
q4h
long acting: 600–1,200 mg q12h
Children 7–12 y: short acting:
100–200 mg q4h
long acting: 600 mg q12h
Children 2–6 y: 50–100 mg q4h
GI upset, drowsiness,
headache, rash, dizziness
Breast-feeding mothers,
pregnancy category C
Not given for prolonged time if
cough persists or accompanied by high fever
Humibid sprinkles may be
swallowed whole or opened
and sprinkled on soft food.
Antitussives
dextromethorphan
(Benylin—15 mg/5 mL) 10 mL q6–8h
(Delsym—30 mg/5 mL)
Adults: 10 mL q12h
Children 2–5 y: 2.5 mL q12h
6–12 y: 5 mL q12h
benzonatate (Tessalon)
100–200 mg tid
Not for ≤ 10 years old
Drowsiness, palpitations,
excitability in children
Drowsiness, headache, GI
upset, confusion
Hypertension, diabetes,
asthma
Pregnancy category C
None
Narcotic Antitussives
codeine phosphate 10 mg, guaifenesin
300 mg tablets
and liquid (Brontex)
Adults: 20 mL q4h
Children 6–12 y: 10 mL q4h
Lightheadedness, dizziness,
sedation, sweating,
nausea, vomiting
Known addiction, cautious
use in asthmatics, COPD,
cardiac disease, seizure
disorders, renal/hepatic
impairment, BPH, head
injuries, hypothyroidism,
and pregnancy
Increased CNS depression if
used with alcohol or other
narcotics
Usually used with antihistamines, expectorants, decongestants, or analgesics
Controlled substance (Drug
Enforcement Agency number
required for prescription)
Phenergan with codeine (codeine 10 mg
and promethazine 6.25 mg/5 mL)
Adults: 5 mL
Children: 2–5 y: 1.25–2.5 mL q4h
6–12 y: 2.5–5 mL q4h
Same as above Same as above Same as above
(continued )
Arcangelo_Chap24.indd 335 10/8/2011 1:52:50 PM
336 UNIT 5 | PHARMACOTHERAPY FOR RESPIRATORY DISORDERS
TABLE 24.2 Overview of Agents for Upper Respiratory Infections (Continued )
Generic (Trade)
Name and Dosage Selected Adverse Events Contraindications Special Considerations
codeine 10 mg, guaifenesin
100 mg/5 mL (Robitussin AC) or
codeine 10 mg, pseudoephedrine
30 mg, and guaifenesin
100 mg/5 mL (Tussar SF)
Adults: 10 mL q4h to maximum of
40 mL/d
Same as above Children: not recommended Same as above
hydrocodone (in combination with other
agents) 5 mg up to 4 qid
Same as above Known addiction; cautious
use in asthmatics, COPD,
cardiac disease, seizure
disorders, renal/hepatic
impairment, BPH, head
injuries, hypothyroidism,
and pregnancy
Same as above
hydrocodone 2.5 mg, guaifenesin
100 mg, pseudoephedrine 30 mg/
5 mL (Duratuss HD)
Same as above Same as above Same as above
Adults: 10 mL q4–6h
Children 6–12 y: 5 mL q2–6h
Maximum of 4 doses/d
hydrocodone 5 mg and homatropine
1.5 mg (Hycodan tablets and syrup)
Same as above Same as above Same as above
Adults: 1 tablet or 5 mL q4–6h
Children: 6–12 y: ½ tablet or 2.5 mL
q4–6h
hydrocodone 5 mg and guaifenesin
100 mg/5 mL (Hycotuss)
Adults: 5 mL after meals and hs
Children: 6–12 y: 2.5–5 mL after
meals and hs
Same as above Same as above Same as above
hydrocodone 10 mg and chlorpheniramine maleate 8 mg/5 mL
(Tussionex)
Adults: 5 mL q12h
Children: 6–12 y: 2.5 mL q12h
Same as above Same as above Same as above
hydrocodone 5 mg guaifenesin 100 mg
per 5 mL (Vicodin Tuss)
Adults: 5 mL at meals and hs
Children 6–12 y: 2.5 mL at meals
and hs
Same as above Same as above Same as above
Combination Products—Non-Narcotic
dextromethorphan hydrobromide
10 mg, brompheniramine maleate
2 mg, pseudoephedrine 30 mg/5 mL
(Bromfed-D, Dimetane-DX)
Adults: 10 mL q4h
Children: 2–5 y: 2.5 mL q4h 6–12 y:
5 mL q4h
Drowsiness, sedation,
nausea, dizziness, palpitations, increased blood
pressure, excitation in
children, constipation
Asthma, lower respiratory
disorders, neonates,
severe hypertension,
severe cardiovascular
disease, within 14 d of
monoamine oxidase inhibitors, nursing mothers; use
cautiously in patients with
history of urinary obstruction, mild hypertension,
and hyperthyroidism
These drugs are combination
antitussives, antihistamines,
and sympathomimetics.
Used for cough and congestion
Pregnancy category C
Not recommended for children
<2 y
These drugs are sold over the
counter.
[Show More]
-2.png)

-3.png)