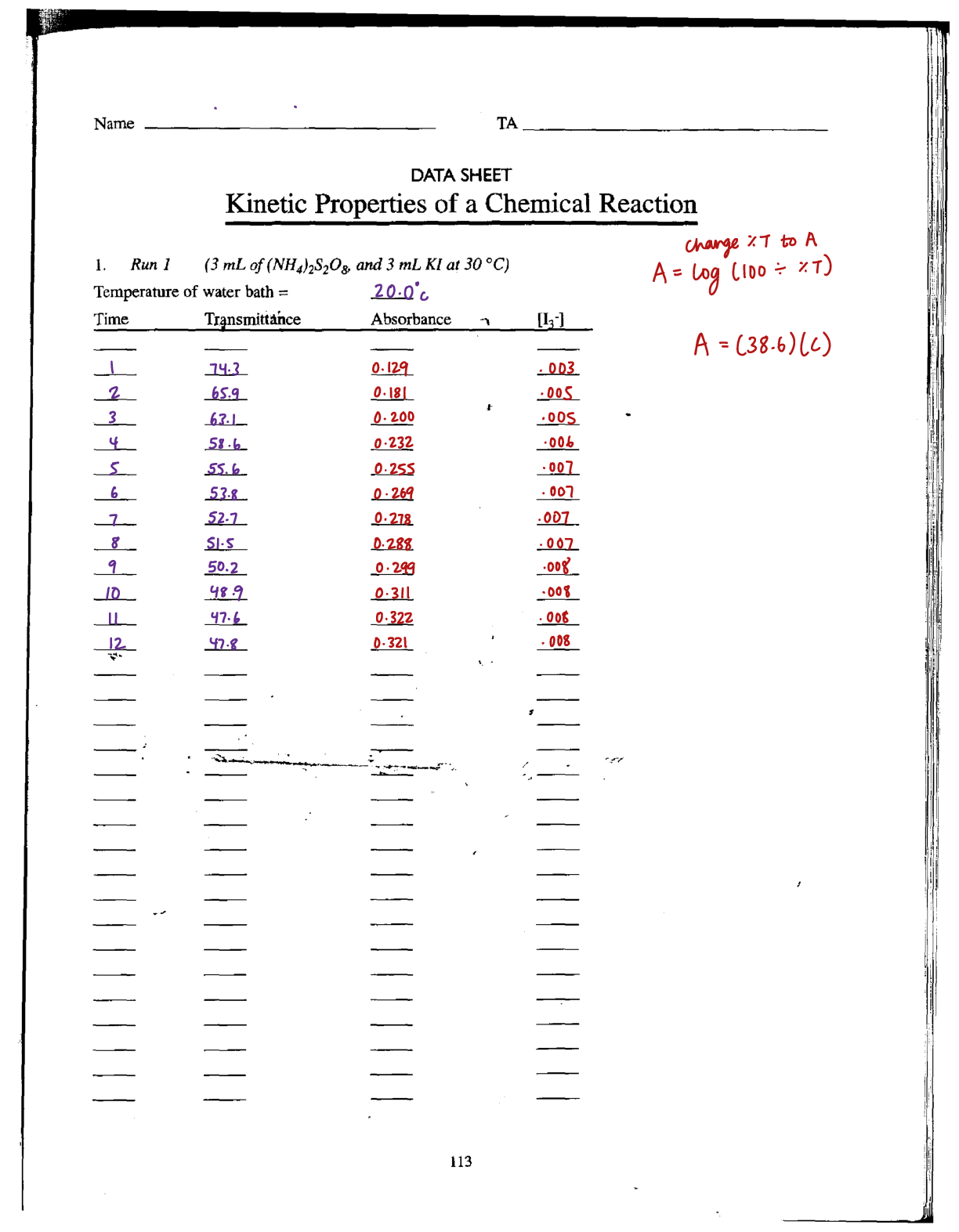
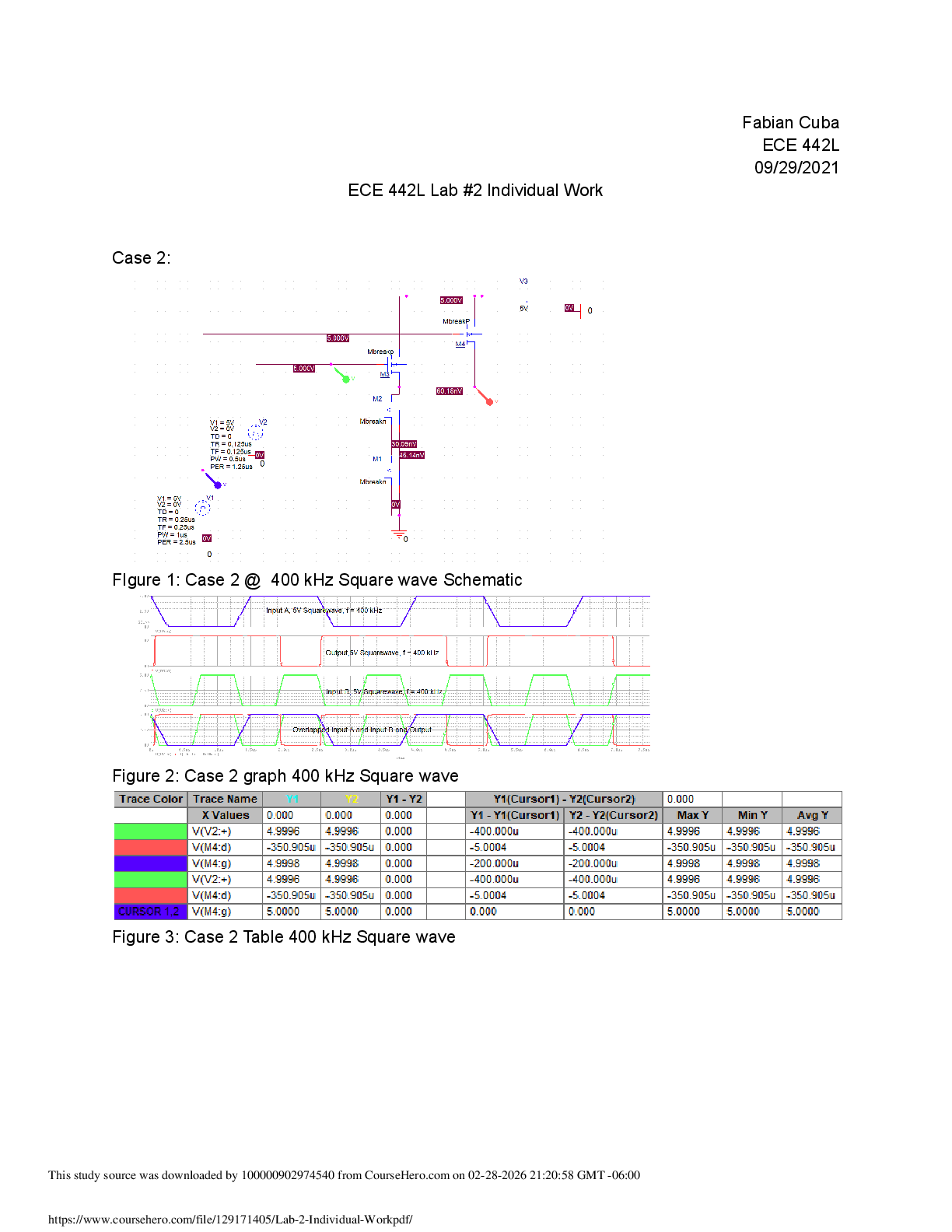
Equilibrium Lab Report
Instructions: For this investigative phenomenon, you will observe the changes that result when
you add more of, or take away some of, one of the components of the equilibrium (known as a
stress)
...
Equilibrium Lab Report
Instructions: For this investigative phenomenon, you will observe the changes that result when
you add more of, or take away some of, one of the components of the equilibrium (known as a
stress) and determine if there are trends in the behavior of equilibrium systems. Record your
observation in the lab report below. You will submit your completed report.!
Title: Equilibrium Lab
Objective(s): Observe color changes that indicate the increasing and decreasing of
concentration in a component of an equilibrium system in order to determine how they
affect the position of equilibrium.
Hypothesis: If the amount of reactants increases, the rate of the forward reaction will
increase to reach a new equilibrium.
Explain the relationship between forward and reverse reactions at equilibrium and predict how
changing the amount of a reactant or product (creating a stress) will affect that relationship.
For example (select one from each underlined section)
If the amount of (reactant or product) increases, the rate of the (forward or reverse) reaction will
(increase or decrease) to reach a new equilibrium.
If the amount of (reactant or product) decreases, the rate of the (forward or reverse) reaction will
(increase or decrease) to reach a new equilibrium.
Procedure:
Access the virtual lab and complete the inquiry experiment. List your controlled variables,
independent variable, and dependent variable. Explain why these are the variables. Summarize
the steps of the experimental procedure.
Materials:
Equilibrium Virtual Lab
Variables:
Remember, controlled variables are factors that remain the same throughout the
experiment. An independent (test) variable changes so that the experimenter can see the
effect on other variables. The dependent (outcome) variable will change in response to
the test variable.
Controlled variables: A, B, C are controlled variables, because they remain the
same throughout the experiment.
Independent variable: The concentration of the components is the independent
variable, because the results of the experiment are determined by this variable.
Dependent variable: The test tubes are the dependent variable, because they are
affected by other variables in the experiment.
Summary of Steps:
Include a summary of steps here. We have included the first two important steps to get
you started. You can summarize the rest from the virtual lab.
1. Write down the equilibrium equation you are investigating using the information
provided by the introduction within the virtual lab activity
2. Select each control test tube to compare the concentrations of reactants and
products. Write down what each indicator color means about the concentrations of
reactants and products in solution.
Pink means:
Blue means:
Purple means:
For each trial, I will add an amount of solution with concentrations equal to or greater than
that of Co(H2O)62+ and CoCl42–. I will add the same amount of solution to Cl–, and record the
initial color of the solution, what is added to the test tube, and the effects of the substance.
A new solution will be added in every trial.
[Show More]











.png)






