Introduction
Enzymes are globular proteins that keep the metabolic pathway flowing (1). Catalysts are the
secondary substance that assists an enzyme when biochemical reaction needs to regulate or accelerate
the rate.
...
Introduction
Enzymes are globular proteins that keep the metabolic pathway flowing (1). Catalysts are the
secondary substance that assists an enzyme when biochemical reaction needs to regulate or accelerate
the rate. When digestive enzymes are produced in the pancreas, they are activated in the small intestine
to prevent the digestive enzyme from destroying the pancreas itself. Another way enzymes influence the
human body is by the clotting factor. When a vessel is damaged the enzyme that promotes blood clot
formation on the wall is catalyzed to prevent any more blood from leaving the broken blood vessel (1).
The enzymes are able to perform their catalytic functions because they take place in a chemical reaction.
A minimum amount of energy designated to begin a chemical reaction is termed activation
energy (2). Every chemical reaction requires a certain amount of this energy. Kinetic energy must be
equal or greater than the activation energy in order for the reaction to perform this is what takes place
with enzyme action. The activation energy is the energy required to break bonds of the reactants in
order for the enzyme to rearrange into the desired mechanism to produce products. Thus, this terms
collision theory in which it states, that molecules involved in a reaction regardless of state or phase will
collide in a particular orientation with an amount of kinetic energy that exceeds the activation energy in
order for the chemical reaction to occur. Kinetic energy in specifics to enzyme action can increase with
temperature, but if the temperature is too high it will denature proteins (1). Hence, when someone has
a high fever it can lead to brain damage. Enzymes are able to regulate chemical reactions by occurring at
normal body temperatures and using a catalyst to decrease the amount of activation energy that is
required (1). If the product of a chemical reaction sits lower than the activation energy then it is an
exothermic reaction, as the reaction releases energy. If the product of a chemical reaction sits higher
than the activation energy than the reaction is endothermic as the energy was absorbed.
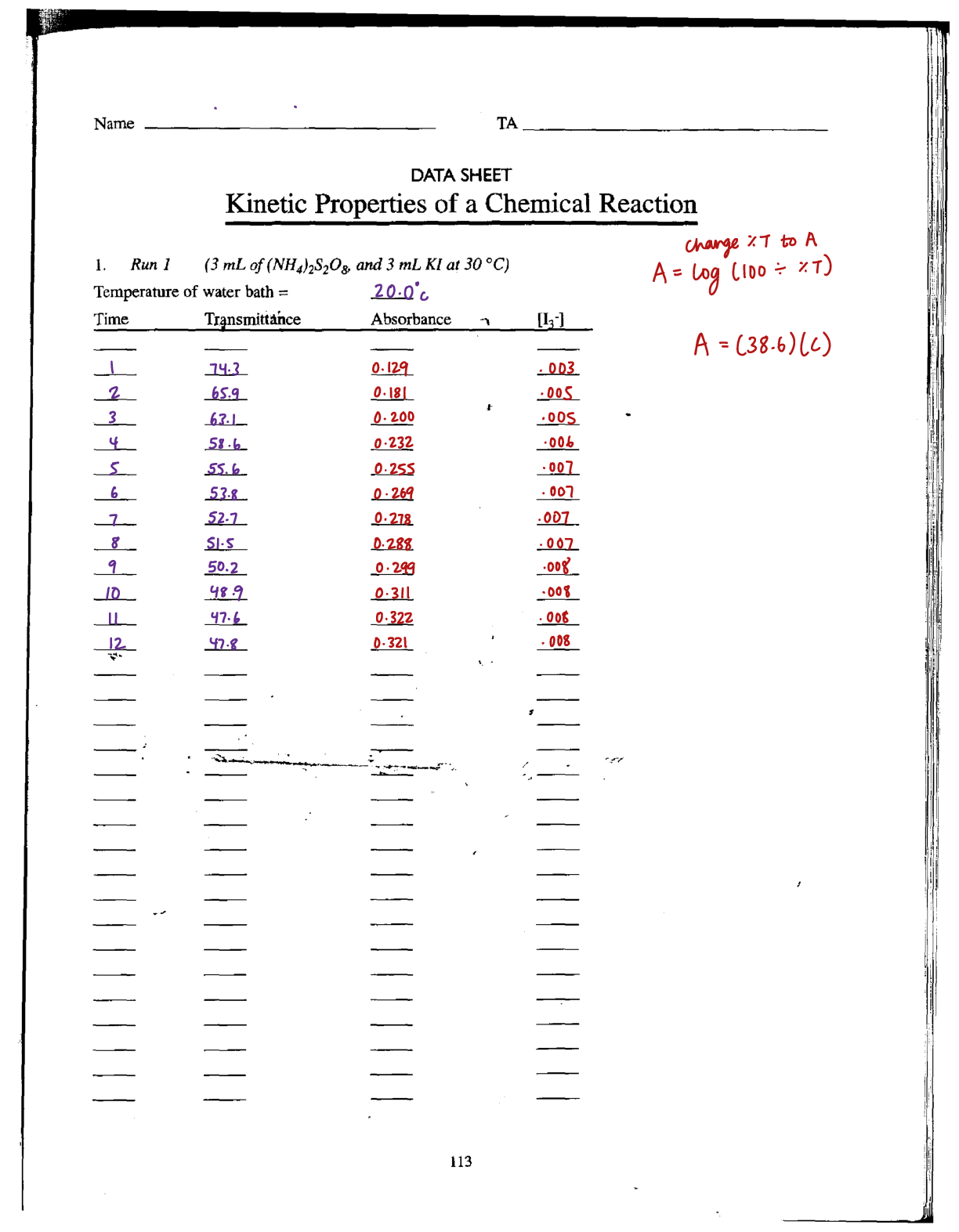
Kinetic energy also refers to the reaction rate, which is the change in concentration of a reactant
or a product with time. Rate laws express the relationship of the reaction rate to the rate constant
through the determination of concentrations of the reactants raised to a certain power. The rate law was
determined by studying the iodide oxidization by hydrogen peroxide this past week, with the following
chemical equation
[Show More]