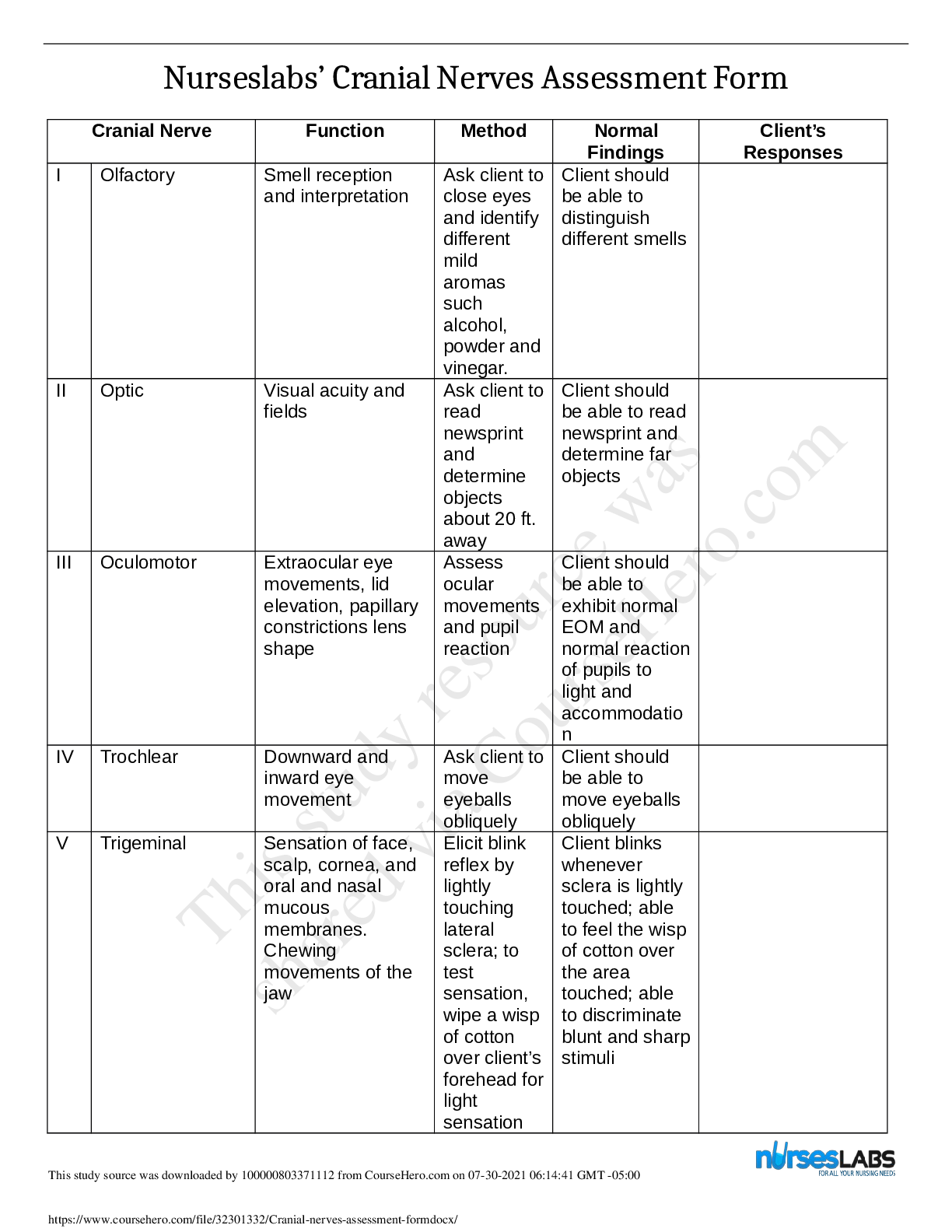
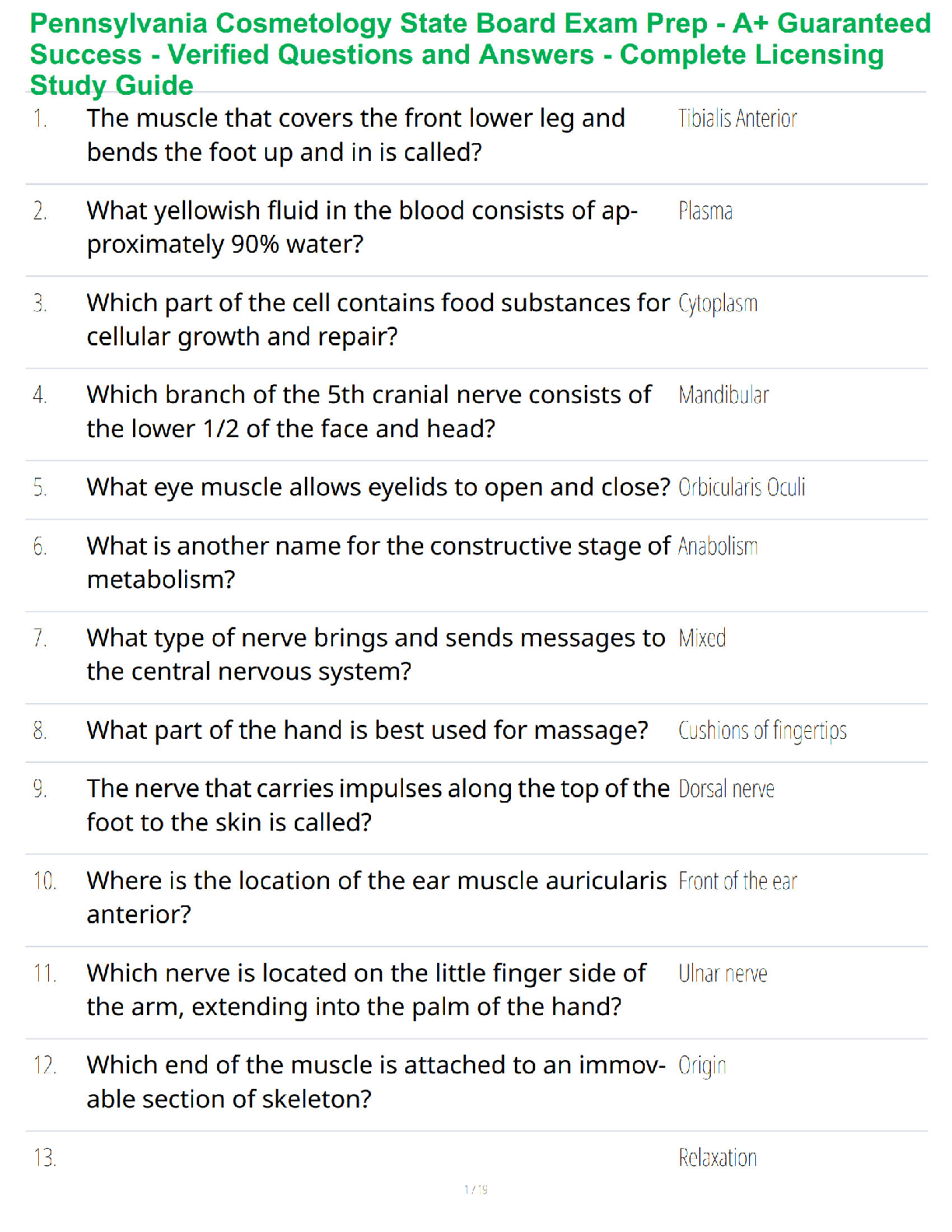
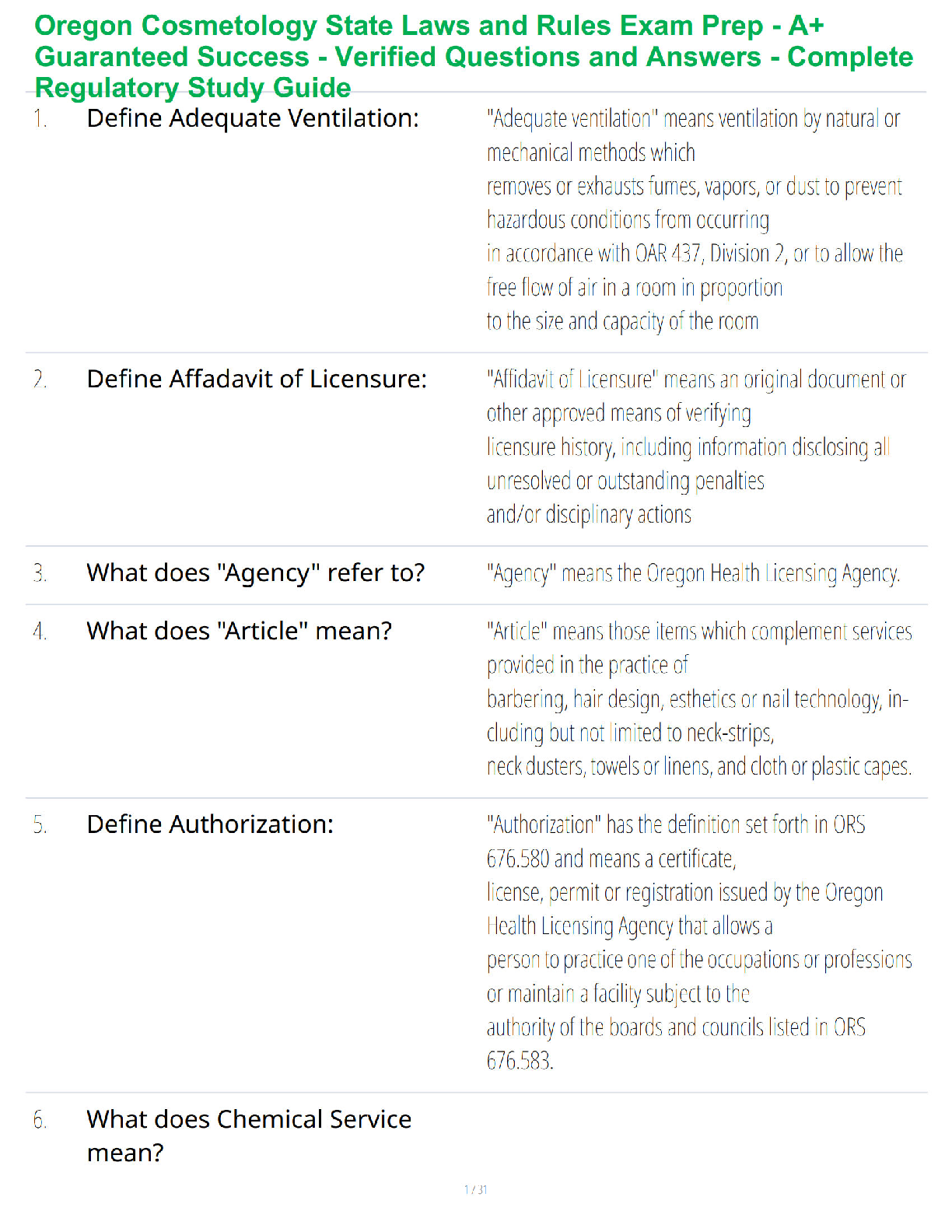
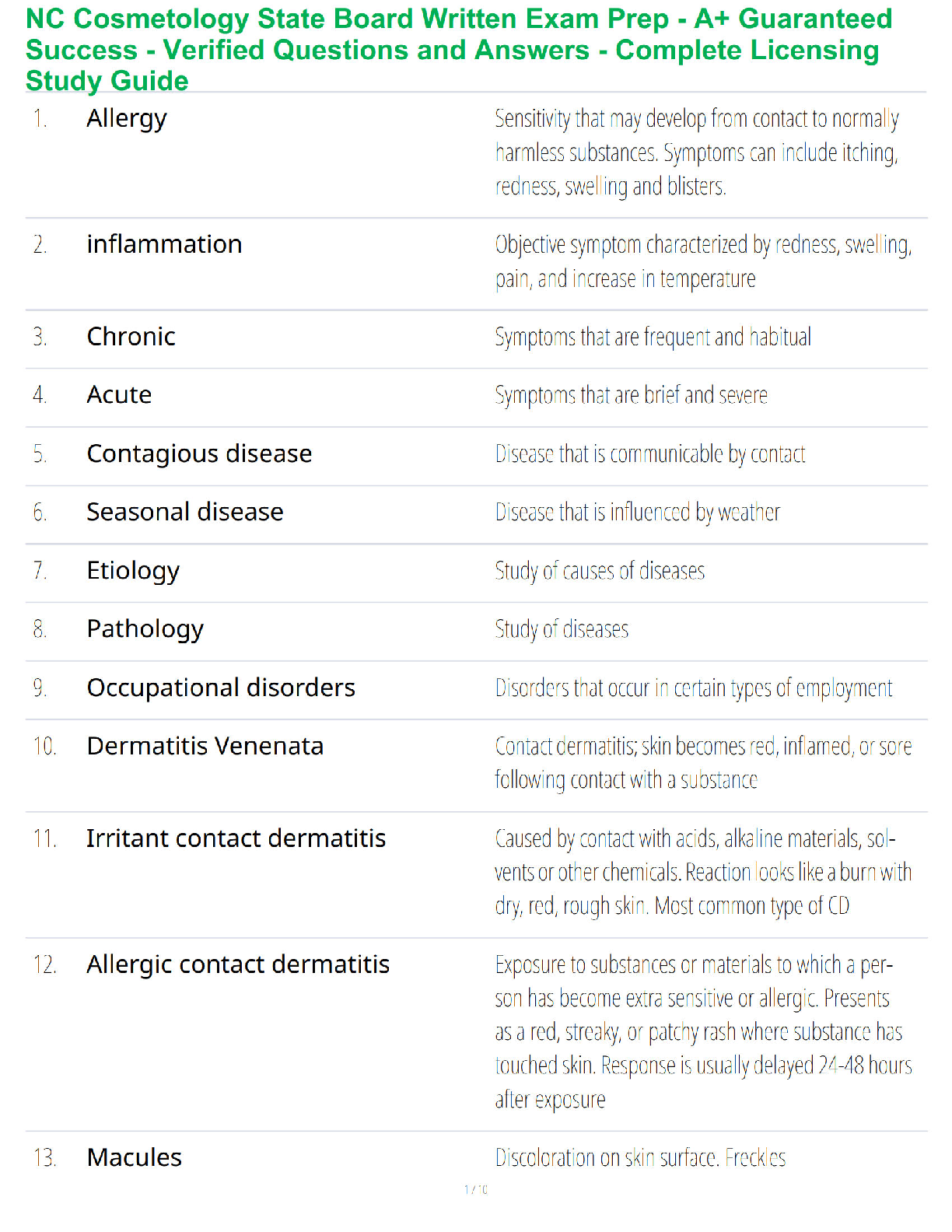
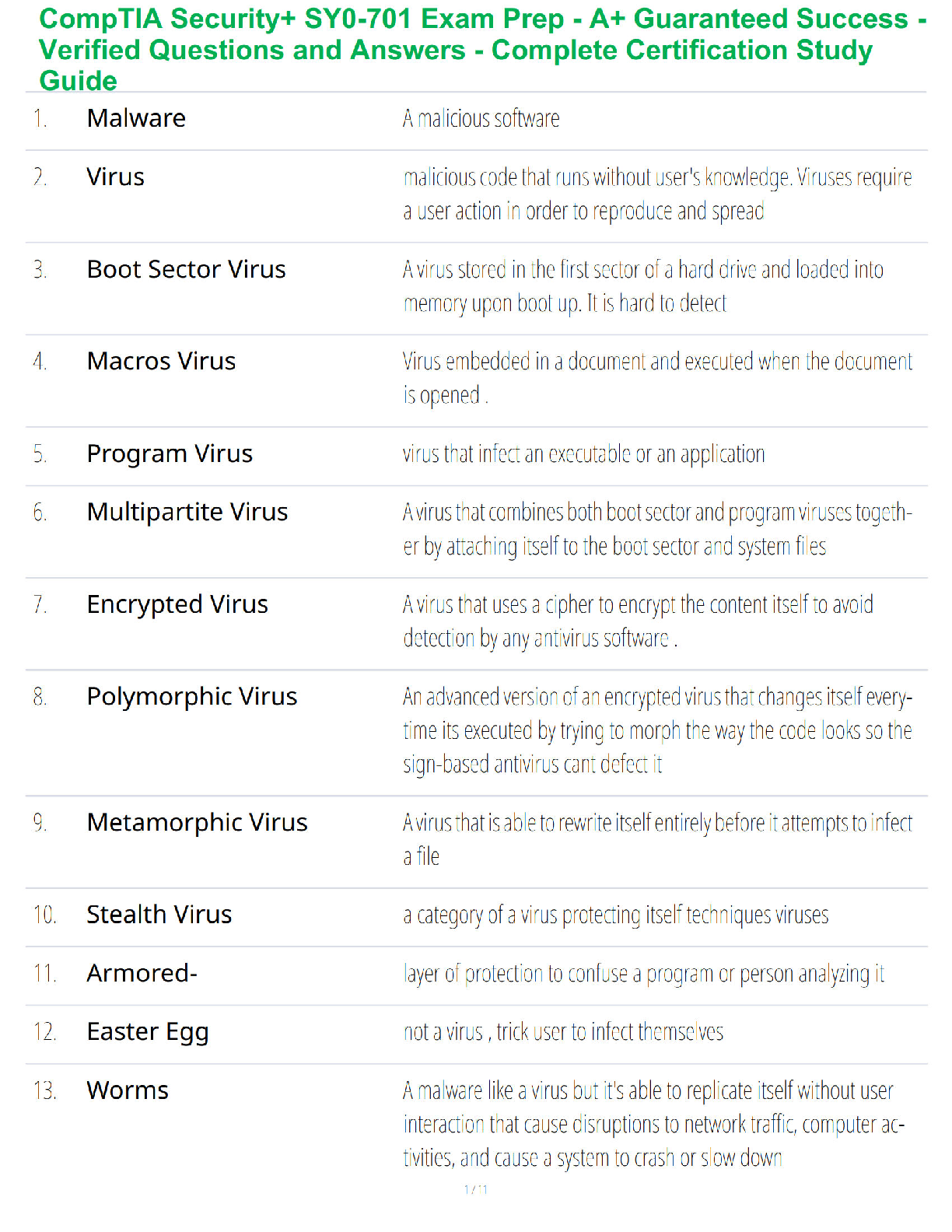
1. Make up a volumetric solution
and carry out a simple acid –
a, d, e, k
Name ________________________
Teacher ______________________
A LEVEL CHEMISTRY LABORATORY
BOOK
This Lab book is for you to evidence your wo
...
1. Make up a volumetric solution
and carry out a simple acid –
a, d, e, k
Name ________________________
Teacher ______________________
A LEVEL CHEMISTRY LABORATORY
BOOK
This Lab book is for you to evidence your work toward standards
Please ensure you use it appropriately;
1. Notes should be recorded as experiments are taking place. They should not be a “neat”
record written at a later date from scraps of paper. However, they should be written
clearly, in legible writing and in language which can be understood by others.
2. Many lab books are used in industry as a source of data, and so should be written in
indelible ink.
3. To ensure that an observer can be confident that all data are included when a lab book
is examined, there should be no blank spaces.
4. Mistakes should be crossed out and re-written. Numbers should not be overwritten,
erased, nor should Tippex be used.
5. Pencil should not be used for anything other than graphs and diagrams.
6. Anything glued in should lie flat and not be folded
7. EACH PAGE SHOULD BE DATED
Compatible withbase titration.
2. Measurement of an enthalpy
change
a, d, k
3. Investigate how the rate of a
reaction changes with
temperature
a, d, k
4. Carry out simple test-tube
reactions to identify cations and
anions in aqueous solution
b, d, k
5. Distillation of a product from a
reaction
b, d, k
6. Testing for alcohols, aldehyde,
alkene and carboxylic acid
b, c, d, k
7. Measuring the rate of reaction:
a) By initial rate method
b) By continuous monitoring
method
a, k, l
8. Measuring the EMF of an
electrochemical cell
j, k
9. Investigate how pH changes
when a weak acid reacts with a
strong base and when a strong
acid reacts with a weak base
a, c, d, f,
k
10. Preparation of:
a) a pure organic solid and test of
its purity
b) a pure organic liquid
a, b, d, g,
h, k
11. Carry out simple test-tube
reactions to identify transition
metal ions in aqueous solution
b, c, d, k
12. Separation of species by
thin-layer chromatography
i, k
[Show More]