MULTIPLE CHOICE
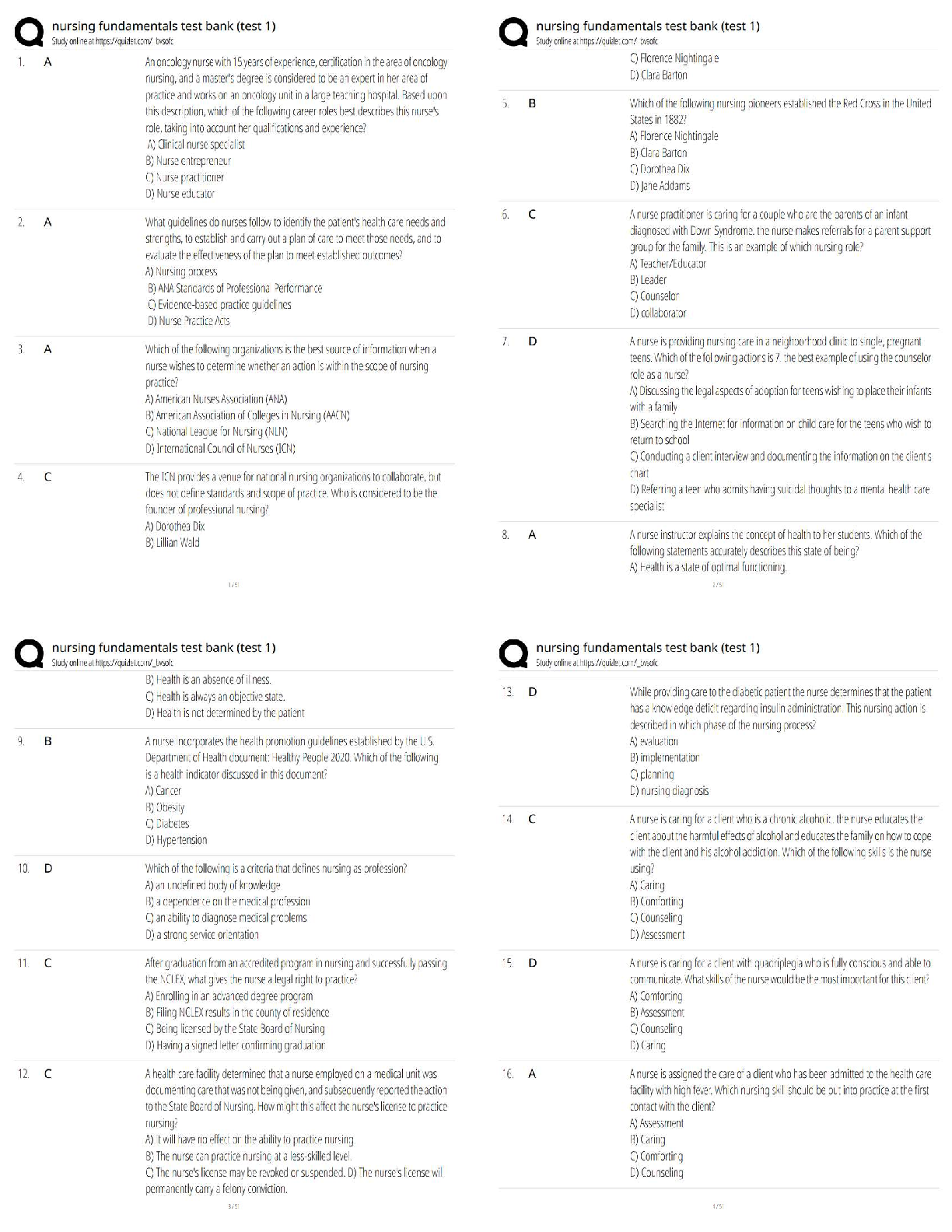
1. Marine organisms contain ____ % water by mass.
a. 40-50
b. 50-60
c. 60-70
d. 70-80
e. 80-90
D PTS: 1 DIF: Recall REF: 67
2. Water can mingle with other elements because it is a:
a.
...
MULTIPLE CHOICE
1. Marine organisms contain ____ % water by mass.
a. 40-50
b. 50-60
c. 60-70
d. 70-80
e. 80-90
D PTS: 1 DIF: Recall REF: 67
2. Water can mingle with other elements because it is a:
a. heating agent.
b. solvent.
c. solid.
d. vapor.
e. nonpolar molecule.
B PTS: 1 DIF: Recall REF: 67
3. The charge of a water molecule is considered:
a. polar.
b. positive.
c. negative.
d. neutral.
e. nonpolar.
A PTS: 1 DIF: Recall REF: 67
4. The polarity of a water molecule is due to:
a. the greater number of hydrogen atoms relative to oxygen.
b. the uneven attraction of electrons to the oxygen atom.
c. the uneven attraction of electrons to the hydrogen atoms.
d. the uneven attraction of protons to the oxygen atom.
e. the number of neutrons in the nucleus.
B PTS: 1 DIF: Recall REF: 67
5. The unique attractive forces that keep molecules of water together are called:
a. strong forces.
b. adhesion.
c. hydrogen bonds.
d. cohesion.
e. weak nuclear forces.
C PTS: 1 DIF: Recall REF: 68
6. Hydrogen bonds are the result of attraction between:
a. the H of one water molecule and the H of another water molecule.
b. the two H atoms of the same water molecule.
c. the H and O of the same water molecule.
d. the H of one water molecule and the O of another water molecule.
D PTS: 1 DIF: Recall REF: 68
7. The property of water whereby molecules tend to stick to one another is called:
a. cohesion.
b. polarity.
c. dissolving ability.
d. adhesion.
e. viscosity.
A PTS: 1 DIF: Recall REF: 69
8. The property of water whereby molecules tend to stick to objects is called:
a. cohesion.
b. surface tension.
c. dissolving ability.
d. adhesion.
e. viscosity.
D PTS: 1 DIF: Recall REF: 69
9. Surface tension allows water molecules to do all the following except:
a. transmit light energy.
b. resist evaporation.
c. form a tight surface layer.
d. support small organisms.
A PTS: 1 DIF: Synthesis REF: 69-70
10. Hydrogen forces have high attractive forces that allow water to have a high:
a. strength.
b. solubility.
c. freezing point.
d. boiling point.
e. rate of evaporation.
D PTS: 1 DIF: Recall REF: 68
11. Water is unique because its solid phase is ____ the liquid phase.
a. denser than
b. similar to
c. less dense than
C PTS: 1 DIF: Recall REF: 68
12. An ion is:
a. a type of gas.
b. an individually charged particle.
c. heavy water.
d. water with an extra hydrogen atom.
e. a neutrally-charged atom.
B PTS: 1 DIF: Recall REF: 69
13. The specific heat of water is
a. a gram of substance.
b. energy required to raise a gram of substance 1 F.
c. calories need to heat seawater.
d. energy required to raise a gram of substance 1 C.
e. energy required to evaporate 1 gram of liquid water.
D PTS: 1 DIF: Recall REF: 69
14. Which low energy light wave length is quickly absorbed by water?
a. Red
b. Orange
c. Yellow
d. Green
e. a, b, and c above
E PTS: 1 DIF: Recall REF: 70
15. Which high energy light wave length can penetrate sea water the deepest?
a. Blue
b. Green
c. Violet
d. Red
e. a, b, and c above
E PTS: 1 DIF: Recall REF: 70
16. In most clear waters, 1% of surface light can penetrate to:
a. 10 m.
b. 20 m.
c. 50 m.
d. 75 m.
e. 100 m.
E PTS: 1 DIF: Recall REF: 70
17. You are SCUBA diving with a friend, who is wearing a red and purple wetsuit. You both descend and conduct your underwater research at 30 m. What color(s) does her wetsuit appear at this depth?
a. black and dark blue.
b. dark blue and purple.
c. dark green and black.
d. dark red and black.
A PTS: 1 DIF: Application REF: 70
18. Light that is absorbed by water is converted to:
a. short wave radiation.
b. long wave radiation.
c. heat.
d. mass.
e. green light.
C PTS: 1 DIF: Recall REF: 70
19. Acids are compounds that:
a. can bind hydrogen ions.
b. can release hydrogen ions.
c. are pH 8 or above.
d. contain hydrogen and hydroxide ions equal in number.
e. raise pH.
B PTS: 1 DIF: Recall REF: 70
20. Bases are compounds that do all the following except
a. they bind hydrogen ions.
b. they release hydrogen ions.
c. they are pH 7 or above.
d. they contain more hydroxide ions than hydrogen ions.
e. they raise pH.
B PTS: 1 DIF: Synthesis REF: 70
21. Water's pH is considered neutral when:
a. hydrogen can be bound.
b. hydrogen can be released.
c. water is slightly alkaline.
d. hydrogen and hydroxide ions are equal in number.
D PTS: 1 DIF: Recall REF: 70
22. The pH scale is a measure of:
a. how many degrees a professor has.
b. the temperature of water.
c. concentration of hydrogen ion in a volume of solution.
d. concentration of hydroxide ions in a volume of solution.
C PTS: 1 DIF: Recall REF: 70
23. The pH of seawater is important to living organisms because:
a. it can affect the functioning of enzymes.
b. it can interfere with metabolism.
c. it can affect growth.
d. All the above.
D PTS: 1 DIF: Synthesis REF: 70–71
24. A trace element's concentration is less than a part per
a. thousand.
b. million.
c. billion.
d. trillion.
e. hundred.
B PTS: 1 DIF: Recall REF: 71
25. Salinity is expressed in parts per
a. hundred.
b. thousand.
c. ten thousand.
d. hundred thousand.
e. million.
B PTS: 1 DIF: Recall REF: 72
26. The average salinity of seawater is ____0/00.
a. 35
b. 37
c. 40
d. 45
e. 50
A PTS: 1 DIF: Recall REF: 72
27. The salinity of seawater at the poles is:
a. high due to evaporation.
b. low due to precipitation.
c. high due to freezing.
d. low due to river input.
e. similar to the open ocean.
C PTS: 1 DIF: Recall REF: 72
28. The salinity of seawater at the equator is:
a. high due to evaporation.
b. low due to precipitation.
c. high due to freezing.
d. low due to river input.
B PTS: 1 DIF: Recall REF: 72
29. Salts are removed from the oceans primarily by
a. absorption by living organisms.
b. removal by sea spray.
c. evaporation.
d. adsorption onto particles.
D PTS: 1 DIF: Recall REF: 72–73
30. When a shallow extension of the sea dries out it leaves salt deposits called:
a. participates.
b. conglomerates.
c. evaporites.
d. rock salt.
e. sedimentary layers.
C PTS: 1 DIF: Recall REF: 72
31. Gases that are regulated by biological processes include:
a. oxygen.
b. carbon dioxide.
c. argon.
d. Both a and b.
e. Both b and c.
D PTS: 1 DIF: Recall REF: 73–74
32. The oxygen minimum layer is found
a. in the ocean trenches.
b. mid-ocean.
c. just below the sunlit surface.
d. at the surface.
e. just above the abyssal plain.
C PTS: 1 DIF: Recall REF: 73
33. In terms of gas solubility in sea water, what is the correct relationship?
a. Carbon dioxide > oxygen > nitrogen
b. Oxygen > carbon dioxide > nitrogen
c. Nitrogen > carbon dioxide > oxygen
d. Carbon dioxide > nitrogen > oxygen
e. Oxygen > nitrogen > carbon dioxide
A PTS: 1 DIF: Recall REF: 73–74
34. To remove carbon dioxide that is dissolved in seawater, you could ________the water sample.
a. chill
b. increase the pressure of
c. increase the salinity of
d. heat
D PTS: 1 DIF: Application REF: 74
35. Seawater is considered buffered due to the presence of:
a. oxygen ions.
b. nitrogen ions.
c. bicarbonate ions.
d. silicon ions.
e. calcium ions.
C PTS: 1 DIF: Recall REF: 74
36. An imbalance of heat received versus heat leaving the earth can lead to:
a. global warming.
b. global cooling.
c. temperature inversion.
d. Both a and b.
e. Both b and c.
D PTS: 1 DIF: Recall REF: 74–75
37. The majority of the heat that is transferred from the earth to the atmosphere is transferred by:
a. radiation of heat into the atmosphere.
b. evaporation of water.
c. absorption by the lithosphere.
d. reflection of heat into space.
B PTS: 1 DIF: Recall REF: 75
38. The following are considered greenhouse gases except:
a. nitrogen.
b. carbon dioxide.
c. methane.
d. chlorofluorocarbons.
A PTS: 1 DIF: Recall REF: 75
39. The mass of a substance divided by its volume is:
a. heat capacity.
b. surface tension.
c. density.
d. salinity.
e. viscosity.
C PTS: 1 DIF: Recall REF: 83
40. The two main factors controlling the density of water are:
a. salinity and temperature.
b. salinity and heat capacity.
c. salinity and pH.
d. pH and heat capacity.
e. temperature and pH.
A PTS: 1 DIF: Recall REF: 83
41. The maximum density of pure water occurs at:
a. -2° C.
b. 0° C.
c. 3° C.
d. 4° C.
e. -4° C.
D PTS: 1 DIF: Recall REF: 83
42. A zone of rapid temperature change is called a(n):
a. pycnocline.
b. thermocline.
c. halocline.
d. isocline.
B PTS: 1 DIF: Recall REF: 83
43. An isopycnal water column is one in which the density ____ with depth.
a. increases
b. decreases
c. remains the same
d. None of the above.
C PTS: 1 DIF: Recall REF: 84
44. The density of air increases with:
a. increasing temperature.
b. decreasing pressure.
c. increasing moisture.
d. decreasing moisture.
D PTS: 1 DIF: Recall REF: 77
45. At 30° north and 30° south, air is:
a. cool and rising.
b. moist and rising.
c. moist and falling.
d. dry and falling.
e. dry and rising.
D PTS: 1 DIF: Recall REF: 79
46. The Coriolis effect is:
a. the downwelling of seawater at the equator.
b. the upwelling of seawater at the equator.
c. the apparent deflection of the path of air and water.
d. the reduction of air speed over the ground as one goes from the equator to the poles.
C PTS: 1 DIF: Recall REF: 78–79
47. A Class 4 hurricane is centered over Bermuda (latitude 32.20 N) and is moving eastwards. Which coastal city on the eastern coast of the United States is in the most peril from its approach?
a. Savannah, GA (latitude 32.08 N)
b. Jacksonville, FL (latitude 30.32 N)
c. Wilmington, NC (latitude 34.23 N)
d. Boston, MA (latitude 42.36 N)
C
Due to Coriolis effect, hurricane will veer to the right as it travels east, making landfall at a higher latitude than where it began.
PTS: 1 DIF: Application REF: 78–79
48. A common generating force for surface waves in the oceans is:
a. wind.
b. gravity.
c. surface tension.
d. capillary action.
e. undersea tectonic activity.
A PTS: 1 DIF: Recall REF: 87–88
49. The designation of winds is referenced from
a. the direction from which they are coming.
b. the direction towards which they are going.
c. folk lore.
d. the need to throw horses overboard.
e. the direction in relation to the observer’s path of travel
A PTS: 1 DIF: Recall REF: 79
50. Deep water waves move through water having a depth that is deeper than ____ the wavelength of the wave.
a.
b.
c.
d.
e.
C PTS: 1 DIF: Recall REF: 88
51. The distance over which wind is blowing is referred to as:
a. fetch.
b. duration.
c. strength.
d. depth.
e. wind span.
A PTS: 1 DIF: Recall REF: 88
52. Gyres are divided into the following types of currents (named below) except
a. Ekman spiral.
b. eastern boundary.
c. western boundary.
d. transverse.
A PTS: 1 DIF: Recall REF: 79–80
53. Tsunamis are usually:
a. deep-water waves.
b. shallow-water waves.
c. wind waves.
d. capillary waves.
e. the result of winter storms.
B PTS: 1 DIF: Recall REF: 89
54. A tidal pattern of two equal high and low tides per day is called a:
a. mixed semidiurnal tide.
b. semidiurnal tide.
c. diurnal tide.
d. spring tide.
e. neap tide.
B PTS: 1 DIF: Recall REF: 91
55. You are on the beach at Corpus Christi, Texas. High tide falls at noon. When will be the next high tide?
a. 6 pm that evening.
b. 12 hours later, at midnight.
c. 6 am the next morning.
d. noon the next day.
D PTS: 1 DIF: Application REF: 91–92
56. A falling tide is called a(n):
a. flood tide.
b. spring tide.
c. neap tide.
d. ebb tide.
e. mixed tide.
D PTS: 1 DIF: Recall REF: 91
TRUE/FALSE
57. The charges on a water molecule are distributed equally.
F PTS: 1 REF: 68
58. Frozen water is more dense than liquid water.
F PTS: 1 REF: 68
59. The capillary action of water is a result of the adhesive properties of water.
T PTS: 1 REF: 69
60. The thermal capacity of water is due to the strong covalent bonds between the hydrogen and oxygen of a water molecule.
F PTS: 1 REF: 69
61. The surface tension of water is a result of the cohesive properties of water.
T PTS: 1 REF: 69
62. On the pH scale, numbers below 7 indicate acidic solutions.
T PTS: 1 REF: 70
63. Trace elements dissolved in seawater are present in concentrations less than one part per thousand.
F PTS: 1 REF: 71
64. Trace elements are not important elements for living organisms.
F PTS: 1 REF: 71
65. The salinity of the world's oceans is increasing as more salts dissolve in the water.
F PTS: 1 REF: 72
66. The concentration of CO2 in the atmosphere is lower than its concentration in the oceans.
T PTS: 1 REF: 73
67. Surface temperature is more important than salinity in determining density of surface waters in coastal areas.
F PTS: 1 REF: 83–84
68. Oceanic surface currents are driven by winds.
T PTS: 1 REF: 79
69. Air in the northern hemisphere is deflected to the left of its direction of movement due to the Coriolis effect.
F PTS: 1 REF: 78
70. Areas between wind convection cells are characterized by having stable and consistent winds.
F PTS: 1 REF: 79
71. Capillary waves are small.
T PTS: 1 REF: 88
72. Plunging waves typically occur on very gently sloping coastlines.
F PTS: 1 REF: 88
73. Tsunamis are the result of the storm surge of hurricanes.
F PTS: 1 REF: 89
74. As a wave approaches shallow water, its speed is reduced.
T PTS: 1 REF: 88–89
MATCHING
Match the gas with the most closely associated atmospheric percentage.
a. 20.99%
b. 0.03%
c. 78.08%
75. Nitrogen
76. Oxygen
77. Carbon dioxide
75. C PTS: 1 REF: 73
76. A PTS: 1 REF: 73
77. B PTS: 1 REF: 73
Match the Percentage by Volume in the Ocean with the gas.
a. 83%
b. 11%
c. 6%
78. Nitrogen
79. Oxygen
80. Carbon dioxide
78. B PTS: 1 REF: 73
79. C PTS: 1 REF: 73
80. A PTS: 1 REF: 73
Match the value of water with its property.
a. 1.00 g/cm3
b. 0 C
c. 100 C
81. Boiling Point
82. Freezing Point
83. Density (at 4 C)
81. C PTS: 1 REF: 68
82. B PTS: 1 REF: 68
83. A PTS: 1 REF: 68
Match the g/kg of seawater with the proper ion.
a. 10.76
b. 2.71
c. 19.35
84. Chloride
85. Sodium
86. Sulfate
84. C PTS: 1 REF: 71
85. A PTS: 1 REF: 71
86. B PTS: 1 REF: 71
Match the pH value with it is most closely associated term.
a. 8
b. 7
c. 6
87. Acidic
88. Basic
89. Neutral
87. C PTS: 1 REF: 70
88. A PTS: 1 REF: 70
89. B PTS: 1 REF: 70
Match the solution with the most closely associated pH.
a. 11
b. 3
c. 8
d. 7
90. Vinegar
91. Seawater
92. Liquid soap
93. Human saliva
90. B PTS: 1 REF: 70
91. C PTS: 1 REF: 70
92. A PTS: 1 REF: 70
93. D PTS: 1 REF: 70
Match the current with the most closely associated flow direction.
a. West to East
b. North to South
c. South to North
d. East to West
94. Gulf Stream
95. Transverse current
96. Eastern-boundary current
97. Equatorial Counter Current
94. C PTS: 1 REF: 80
95. A PTS: 1 REF: 80
96. B PTS: 1 REF: 80
97. D PTS: 1 REF: 80
Match the 24 hr tidal cycle with its most closely associated term.
a. one low and high
b. two different highs and lows
c. two equal highs and lows
98. Semidiurnal
99. Diurnal
100. Mixed tide
98. C PTS: 1 REF: 91–92
99. A PTS: 1 REF: 91–92
100. B PTS: 1 REF: 91–92
Match the wave term with the most closely associated description.
a. a wave that bends
b. water depth < a wave's length
c. water depth > a wave's length
101. Deepwater wave
102. Shallowater wave
103. Wave refraction
101. C PTS: 1 REF: 88
102. B PTS: 1 REF: 88
103. A PTS: 1 REF: 88
Match the wave type with the most closely associated description.
a. no longer influenced by wind from a storm
b. wind generated and restored by gravity
c. increasing in size due to winds from a storm
104. Progressive wave
105. Forced wave
106. Free wave
104. B PTS: 1 REF: 88
105. C PTS: 1 REF: 88
106. A PTS: 1 REF: 88
ESSAY
107. Describe four properties of water that make it indispensable for life as we know it on Earth. How do the properties of water arise?
108. What are hydrogen bonds?
109. What property of water allows it to stick to objects and make them wet?
110. What is capillary action of water and how does it occur?
111. Large bodies of water maintain relatively constant temperatures compared to terrestrial locations. Why is this so, and how is this feature of water important for life?
112. Define the term "evaporite".
113. List the most abundant ions in seawater, from most to least abundant.
114. Which of the two gases, oxygen or carbon dioxide, is present in the oceans in much greater proportions than in air? Why might this be the case?
115. Describe the physical factors involved in the layering of the oceans.
116. What are atmospheric low pressure zones? Where are they found, and why are these zones significant for traditional marine navigation?
117. Explain what happens at the molecular level when a salt such as NaCl dissolves in water.
118. Even though seawater continues to dissolve minerals from rocks and new minerals are added from the interior of the earth, the salinity of the oceans is not increasing over time. Explain 3 reasons that cause this.
119. Tropical areas absorb more heat than they lose and polar areas lose more heat than they gain. How is it that the tropics do not continue to get warmer and the poles don't get colder with time?
120. The oceans are thought to regulate temperatures of the whole planet. Explain this process in terms of the heat capacity of water.
121. What 2 factors can cause a stratified water column to become unstable and to mix?
122. Explain why thermohaline circulation (the vertical mixing of the water column due to density differences) is important for living organisms.
123. Describe the astronomical conditions that give rise to the extreme spring tides during certain periods of the month.
[Show More]












.png)
.png)
.png)